Team:UNIPV-Pavia/Notebook/Week15
From 2008.igem.org
Notebook
| Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | Week 6 | Week 7 |
|---|---|---|---|---|---|---|
| Week 8 | Week 9 | Week 10 | Week 11 | Week 12 | Week 13 | Week 14 |
| Week 15 | Week 16 | Week 17 | Week 18 | Week 19 | Week 20 | Week 21 |
| Week 22 | Week 23 | Week 24 |
Week 15: 08/25/08 - 08/29/08
08/25/08
- Plasmid digestion for:
- R0051 (S-P)
- R0040 (S-P)
- B0030-C0061-B1006-R0062-B0030-E0040-B1006 (E-X) (=Lig.b (E-X))
- B1006 (E-X)
- Lig.12 (E-S)
- Run/gel extraction.
- Ligations:
- BBa_R0051 (S-P) - BBa_E0240 (X-P) (for promoter test)
- BBa_R0040 (S-P) - BBa_E0240 (X-P) (for promoter test)
- Lig.12 (E-S) - B0030-C0061-B1006-R0062-B0030-E0040-B1006 (E-X) (for AND logic gate test)
- Lig.12 (E-S) - BBa_B1006 (E-X) (to re-perform mutated assemblies)
- We incubated ligations at 16°C overnight.
- We ordered 3OC6HSL (Sigma).
08/26/08
- We transformed/plated ligations.
08/27/08
- Plates grew correctly. We checked colony fluorescence of three plates under UV rays:
- R0051-E0240 glowed
- R0040-E0240 glowed
- Lig.12-Lig.b (R0051-B0030-C0062-B0030-C0061-B1006-R0062-B0030-E0040-B1006) glowed
- We picked up fluorescent colonies from these three plates to infect three 15 ml falcon tubes containing 1 ml of LB + Amp. We let the culture grow at 37°C, 220 rpm for 3 hours, then we prepared three 50 ul samples and watch them at microscope. Results are shown in The Project section (Experiments).
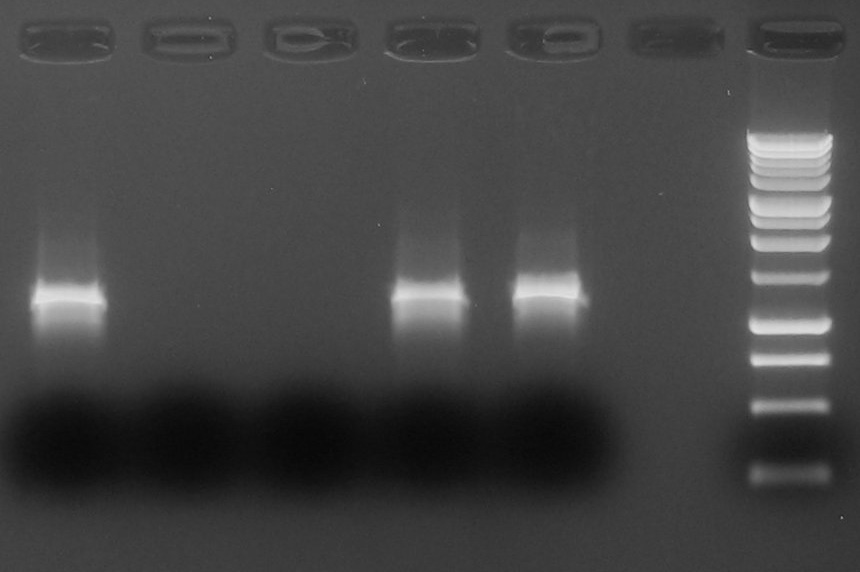
- Colony PCR for 5 colonies of Lig.12-B1006 (called Lig.22).
- Gel results: OK! we chose 1st colony to grow a 9 ml overnight culture.
08/28/08
- Glycerol stocks/miniprep for Lig.22.
- We sent purified plasmids to Primm for sequencing.
- We infected 9 ml of LB + Amp with 30 µl of R0062 glycerol stock.
08/29/08
- Glycerol stock/miniprep for R0062.
 "
"