Team:Paris/August 13
From 2008.igem.org
(Difference between revisions)
(→Minipreps: Plasmid extraction) |
(→Minipreps: Plasmid extraction) |
||
| Line 74: | Line 74: | ||
|L100.1 | |L100.1 | ||
| rowspan="3"| rbs TetR - ECFP<br>D110 (BV) - D130 (BI) | | rowspan="3"| rbs TetR - ECFP<br>D110 (BV) - D130 (BI) | ||
| - | | rowspan="3"| [[Image:Part_icon_rbs.png]][[Image:Icon_coding.png]][[Image:Part_icon_rbs.png]][[Image:Part_icon_reporter.png]][[Image:Part_icon_terminator.png]] | + | | rowspan="3"| [[Image:Part_icon_rbs.png]][[Image:Icon_coding.png]][[Image:Part_icon_rbs.png]][[Image:Part_icon_reporter.png]][[Image:Part_icon_terminator.png]][[Image:Part_icon_terminator.png]] |
|- | |- | ||
|MP147.2 | |MP147.2 | ||
| Line 85: | Line 85: | ||
|L101.1 | |L101.1 | ||
| rowspan="3"| rbs TetR - GFP tripart<br>D110 (BV) - D131 (BI) | | rowspan="3"| rbs TetR - GFP tripart<br>D110 (BV) - D131 (BI) | ||
| - | | rowspan="3"| [[Image:Part_icon_rbs.png]][[Image:Icon_coding.png]][[Image:Part_icon_rbs.png]][[Image:Part_icon_reporter.png]][[Image:Part_icon_terminator.png]] | + | | rowspan="3"| [[Image:Part_icon_rbs.png]][[Image:Icon_coding.png]][[Image:Part_icon_rbs.png]][[Image:Part_icon_reporter.png]][[Image:Part_icon_terminator.png]][[Image:Part_icon_terminator.png]] |
|- | |- | ||
|MP148.2 | |MP148.2 | ||
| Line 96: | Line 96: | ||
|L114.1 | |L114.1 | ||
| rowspan="3"| AracpBAD - gfp tripart<br>D126 (BV) - D131 (BI) | | rowspan="3"| AracpBAD - gfp tripart<br>D126 (BV) - D131 (BI) | ||
| - | | rowspan="3"| [[Image:Part_icon_regulatory.png]][[Image:Part_icon_rbs.png]][[Image:Part_icon_reporter.png]][[Image:Part_icon_terminator.png]] | + | | rowspan="3"| [[Image:Part_icon_regulatory.png]][[Image:Part_icon_rbs.png]][[Image:Part_icon_reporter.png]][[Image:Part_icon_terminator.png]][[Image:Part_icon_terminator.png]] |
|- | |- | ||
|MP149.2 | |MP149.2 | ||
| Line 107: | Line 107: | ||
|L120.1 | |L120.1 | ||
| rowspan="3"| tetR repressible promoter - ECFP<br>D106 (BV) - D130 (BI) | | rowspan="3"| tetR repressible promoter - ECFP<br>D106 (BV) - D130 (BI) | ||
| - | | rowspan="3"| [[Image:Part_icon_regulatory.png]][[Image:Part_icon_rbs.png]][[Image:Part_icon_reporter.png]][[Image:Part_icon_terminator.png]] | + | | rowspan="3"| [[Image:Part_icon_regulatory.png]][[Image:Part_icon_rbs.png]][[Image:Part_icon_reporter.png]][[Image:Part_icon_terminator.png]][[Image:Part_icon_terminator.png]] |
|- | |- | ||
|MP150.2 | |MP150.2 | ||
| Line 118: | Line 118: | ||
|L122.1 | |L122.1 | ||
| RBS-lasI - ECFP <br>D107 (BV) - D130 (BI) | | RBS-lasI - ECFP <br>D107 (BV) - D130 (BI) | ||
| - | | [[Image:Part_icon_rbs.png]][[Image:Icon_coding.png]][[Image:Part_icon_rbs.png]][[Image:Part_icon_reporter.png]][[Image:Part_icon_terminator.png]] | + | | [[Image:Part_icon_rbs.png]][[Image:Icon_coding.png]][[Image:Part_icon_rbs.png]][[Image:Part_icon_reporter.png]][[Image:Part_icon_terminator.png]][[Image:Part_icon_terminator.png]] |
|- | |- | ||
|MP152.1 | |MP152.1 | ||
|L123.1 | |L123.1 | ||
| rowspan="3"| RBS lasI - gfp tripart<br>D107 (BV) - D131 (BI) | | rowspan="3"| RBS lasI - gfp tripart<br>D107 (BV) - D131 (BI) | ||
| - | | rowspan="3"| [[Image:Part_icon_rbs.png]][[Image:Icon_coding.png]][[Image:Part_icon_rbs.png]][[Image:Part_icon_reporter.png]][[Image:Part_icon_terminator.png]] | + | | rowspan="3"| [[Image:Part_icon_rbs.png]][[Image:Icon_coding.png]][[Image:Part_icon_rbs.png]][[Image:Part_icon_reporter.png]][[Image:Part_icon_terminator.png]][[Image:Part_icon_terminator.png]] |
|- | |- | ||
|MP151.2 | |MP151.2 | ||
Revision as of 15:50, 15 August 2008
Sequencing Minipreps we did yesterday
Minipreps: Plasmid extraction
Glycerol Stocks
Assay to purify a PCR product using the Qiaquick Gel Extraction kit
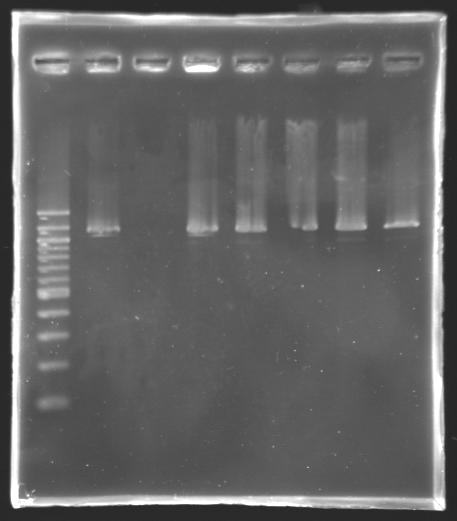
=> see Gel 1, well n° 8 ==> results: PCR products can be purified using the Qiaquick Gel Extraction kit. We just have to replace the buffer PBI by the buffer QG! PCR screening4 more transformants of L130 (pFlhB into J61002) are screened by PCR.
Electrophoresis
==> The L130 transformants analysed are not correct. Transformation of the ligation we did yesterdayWe transformed L 139, L140, L141 and L142 following the standard protocol using Invitrogen's TOP 10 chemically competent cells. The positive control is a transformation with pUC19 and the negative control has no plasmid. Promoter Characterization: work plan |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"