Team:Tokyo Tech
From 2008.igem.org
(→Development of low pressure inducible promoter) |
|||
| Line 162: | Line 162: | ||
<html> | <html> | ||
<body> | <body> | ||
| - | + | <div> | |
| + | It is known that lac promoter is induced under 30 MPa (Kato et al. 1994).However, 30 MPa is too high to use as input. | ||
| + | </div> | ||
| + | <div> | ||
| + | Therefore, we are trying to develop low pressure inducible promoter. | ||
| + | </div> | ||
| + | </body> | ||
| + | </html> | ||
| - | + | == Method == | |
| - | + | === Principle === | |
| - | + | LacI binds to lacI binding site and repress lac promoter. | |
| - | + | In addition, pressure of 30 MPa activated the lac promoter. | |
| - | + | We propose a hypothesis that 30 MPa induce a conformational change in lacI reprssure and change the affinity for lacI binding site weaker. Therefore, lacI can't repress lac promoter under 30 MPa. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | === Strategy === | |
| + | <gallery widths="360px" heights="190px" > | ||
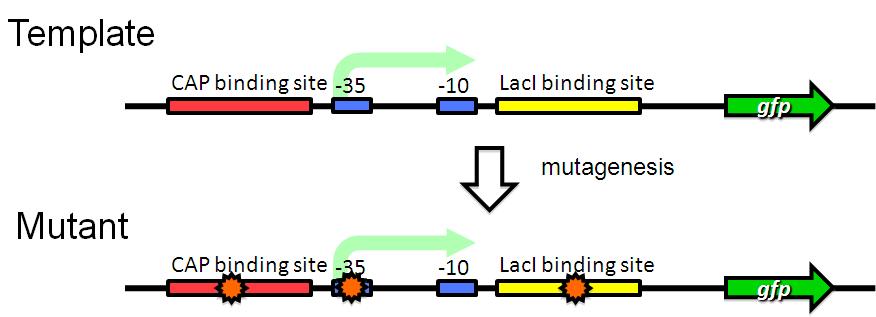
| + | Image:Tech dev 1.JPG|Figure 1 - RCP random mutagenesis | ||
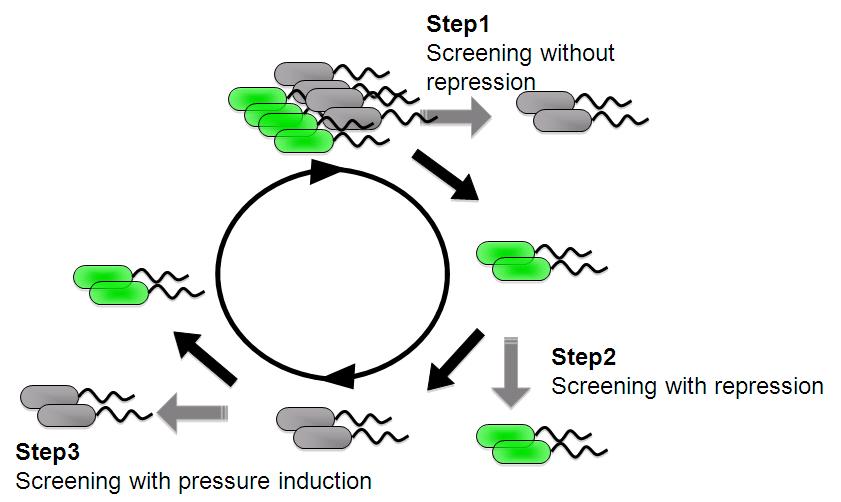
| + | Image:Tech dev 2.JPG|Figure 2 - Strategy of sorting with FACS | ||
| + | </gallery> | ||
| - | + | We are trying to develop low pressure inducible promoter by PCR random mutagenesis to lac promoter in order to reduce affitity to lacI. | |
| - | + | And we are screening an E. coli library for promoters that are induced under low pressure using a fluorescence activated cell sorter (FACS). | |
| - | + | This scheme is based on the ability to separate bacteria with a FACS in response to expression, or lack of expression, of a fluorescent marker. | |
| - | + | *'''step 1''' - Fluorescent bacteria without repressor protein were collected by FACS. This sorted pool contains bacteria bearing both constitutive and low pressure-inducible promoter. | |
| - | + | ||
| - | < | + | *'''step 2''' - Constitutive promoter are removed with repressor protein and sorting all non-fluorescent bacteria. |
| - | < | + | |
| + | *'''step 3''' - A final passage through under pressure with repressor protain and sorting for fluorescent bacteria removes false negatives and enriches for bacteria bearing promoter that are low pressure inducible. | ||
| + | |||
| + | == Results== | ||
| + | We finished step 1. | ||
| + | Fluorescent and non-fluorescent bacteria were sorted and we characterized their promoter. | ||
| + | === Sequence and Characterization=== | ||
| + | <gallery widths="430px" heights="220px" > | ||
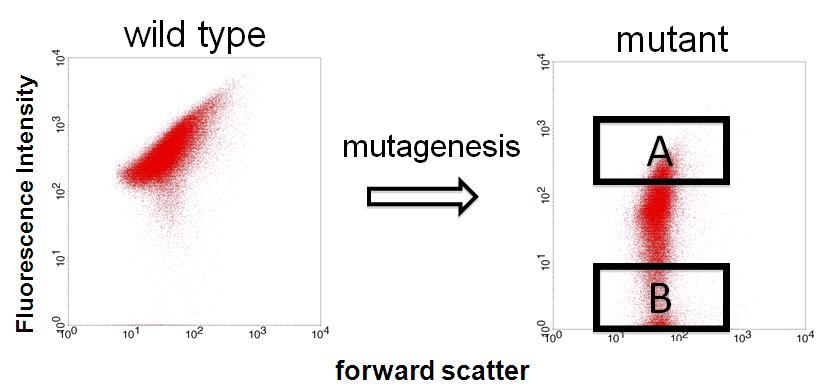
| + | Image:Dev 3.JPG|Figure 3 - FACS | ||
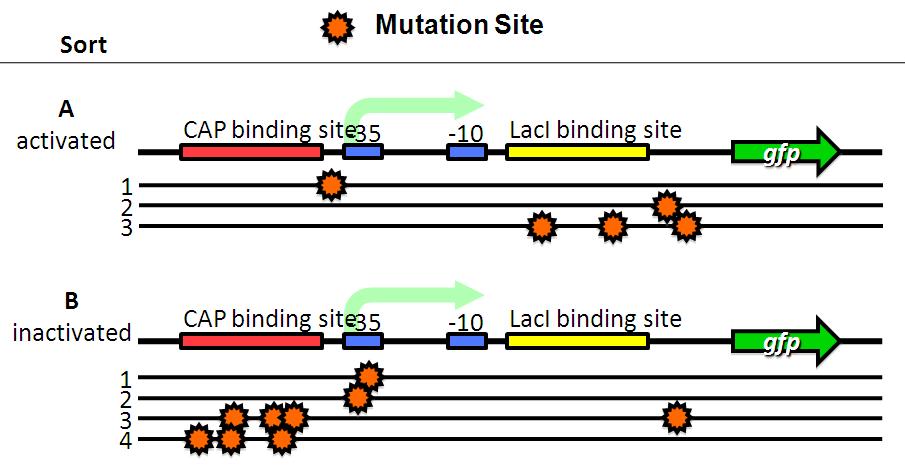
| + | Image:Tech dev 4.JPG|Figure 4 - Sequensing | ||
| + | </gallery> | ||
| + | We sorted fluorescent (A) and non-fluorescent bacteria (B) with a FACS. | ||
| + | Then, we analyze these base sequences. | ||
| + | *A have mutations in lacI binding site or non-functional DNA. | ||
| + | *B have mutations in CAP binding site, -35 or non-functional DNA. | ||
| + | |||
| + | == Conclusion== | ||
| + | We have successfully demonstrated that it is possible to collect objective promoter by PCR random mutagenesis and screening with a FACS. | ||
| + | So we clearly believe that we can screen low pressure inducible lac promoter mutant with this strategy. | ||
=== <font size=5>'''Genetic toggle switch to implement rewritable function'''</font> === | === <font size=5>'''Genetic toggle switch to implement rewritable function'''</font> === | ||
Revision as of 14:28, 26 October 2008
| Home | Construction | Acrylic container | Development of promoter | Genetic toggle switch | Parts Submitted to the Registry | Result | Our Team | Acknowledgements |
|---|
Contents |
Our project
|
We want to make a Coli Touch!!
To make a Coli Touch, we use input style of pressure. |
 |
|
Why pressure?
First, there were various ways as input.
For example, small molecule, heat, and light were used until iGEM 2007.
But no one used pressure as input.
Moreover, past way(small molecule, heat and light) are difficult to induct uniformly.
Pressurize can induct unifomly.It's prospect of technological application in confirmatory experiment. |
|
We use pressure sensitive parts.
One paper says "One promoter is sensitive to pressure.". So we focused on it. |
Acrylic container
 |
|
We create devices for confirming pressure response of lac promoter. This is the first step of creating touch display. This device made of Acrylic glasses and has two holes (show figure). Each hole contains tubes and water. Inside tubes E. coli is cultivated. Pressure can travel to inside tubes. One hole (A) is covered with a plastic tape (show figure). Therefore the hole is pressurized.The other (B) is covered with a block made of an acrylic glass. (show figure) Therefore the hole is not pressurized by water. |
|
E.coli type in tubes |
 |
* “Ptet” on pSB6 plasmid (E.coli strain; JM109) |
|
After pressurized the container, we observed the E.coli by a fluorescence microscope. The result shows below. |
 |
 |
Aplication project
|
About one paper, 94 times activity was observed at 30MPa for the lac
promoter compared with the level expressed at atmospheric pressure.
(cf.Takako Sato, Chiaki Kato,and Koki Horikoshi(1994) Effect of high
pressure on gene expression by lac and tac promoters in Escherichia
coli.Marine Biotechnology 3:89-92)
|
Development of low pressure inducible promoter
Method
Principle
LacI binds to lacI binding site and repress lac promoter. In addition, pressure of 30 MPa activated the lac promoter. We propose a hypothesis that 30 MPa induce a conformational change in lacI reprssure and change the affinity for lacI binding site weaker. Therefore, lacI can't repress lac promoter under 30 MPa.
Strategy
We are trying to develop low pressure inducible promoter by PCR random mutagenesis to lac promoter in order to reduce affitity to lacI. And we are screening an E. coli library for promoters that are induced under low pressure using a fluorescence activated cell sorter (FACS). This scheme is based on the ability to separate bacteria with a FACS in response to expression, or lack of expression, of a fluorescent marker.
- step 1 - Fluorescent bacteria without repressor protein were collected by FACS. This sorted pool contains bacteria bearing both constitutive and low pressure-inducible promoter.
- step 2 - Constitutive promoter are removed with repressor protein and sorting all non-fluorescent bacteria.
- step 3 - A final passage through under pressure with repressor protain and sorting for fluorescent bacteria removes false negatives and enriches for bacteria bearing promoter that are low pressure inducible.
Results
We finished step 1. Fluorescent and non-fluorescent bacteria were sorted and we characterized their promoter.
Sequence and Characterization
We sorted fluorescent (A) and non-fluorescent bacteria (B) with a FACS. Then, we analyze these base sequences.
- A have mutations in lacI binding site or non-functional DNA.
- B have mutations in CAP binding site, -35 or non-functional DNA.
Conclusion
We have successfully demonstrated that it is possible to collect objective promoter by PCR random mutagenesis and screening with a FACS. So we clearly believe that we can screen low pressure inducible lac promoter mutant with this strategy.
Genetic toggle switch to implement rewritable function
|
A touch display are required a function of rewritable. Rewritable function consists of writable and erasable one. We adopted a genetic toggle switch that responds to pressure (show figure) |

 "
"