Team:University of Lethbridge/Notebook/Project2August
From 2008.igem.org
m |
m |
||
| Line 43: | Line 43: | ||
====Roxanne==== | ====Roxanne==== | ||
-Ran the PCR Product on a 3% gel using only 1uL of DNA from the riboswitch. | -Ran the PCR Product on a 3% gel using only 1uL of DNA from the riboswitch. | ||
| + | |||
| + | ===August 16, 2008=== | ||
| + | ====Nathan Puhl, Roxanne, Munima==== | ||
| + | -Restriction Digested the purified riboswitch and pSB1A7 with XbaI and SpeI, let it run for 4 hours | ||
| + | |||
| + | ====Nathan Puhl, Roxanne==== | ||
| + | -Ran all of the restricted pSB1A7 plasmid through a 1% agarose gel at 100V for 25 minutes. | ||
| + | -Ran a gel extraction on the pSB1A7 cut plasmid, and ran a PCR clean-up reaction on the digested RS1 and RS2 amplicons. | ||
| + | -Ran 1 uL of each on a 1% to quantify the amount of DNA present. | ||
| + | -Ligated RS1 + pSB1A7, and RS2 + pSB1A7, using T4 DNA Ligase. | ||
| + | |||
| + | -1 uL of RS1/RS2 | ||
| + | -4 uL of pSB1A7 | ||
| + | -1 uL of 10x T4 DNA Ligase Buffer | ||
| + | -0.33 uL of T4 DNA Ligase | ||
| + | -3.67 uL of water | ||
| + | |||
| + | allowed the reaction to go overnight | ||
| + | |||
| + | |||
| + | ===August 17, 2008=== | ||
| + | ====Nathan Puhl==== | ||
| + | -Transformed DH5a cells with the pSB1A7 + RS1, and pSB1A7 + RS2 plasmids. | ||
| + | |||
| + | -Plated on semi-solid agar plates containing 100 ug/mL of ampicillin. | ||
| + | |||
| + | |||
| + | ===August 18=== | ||
| + | ====Christa, Nathan Puhl, Munima==== | ||
| + | -Nathan checked the plates for growth. Colonies are present. | ||
| + | |||
| + | -Ran a colony PCR of the pSB1A7 + RS1, and pSB1A7 + RS2 recombinant cells transformed by Nathan and Roxanne on August 17. | ||
| + | |||
| + | -Inoculated the cells into tubes of liquid media + 100 ug/ml of ampicillin. | ||
| + | |||
| + | Roxanne will remove the PCR tube from the thermocycler in the morning. | ||
| + | |||
| + | |||
| + | ===August 19, 2008=== | ||
| + | ====Roxanne==== | ||
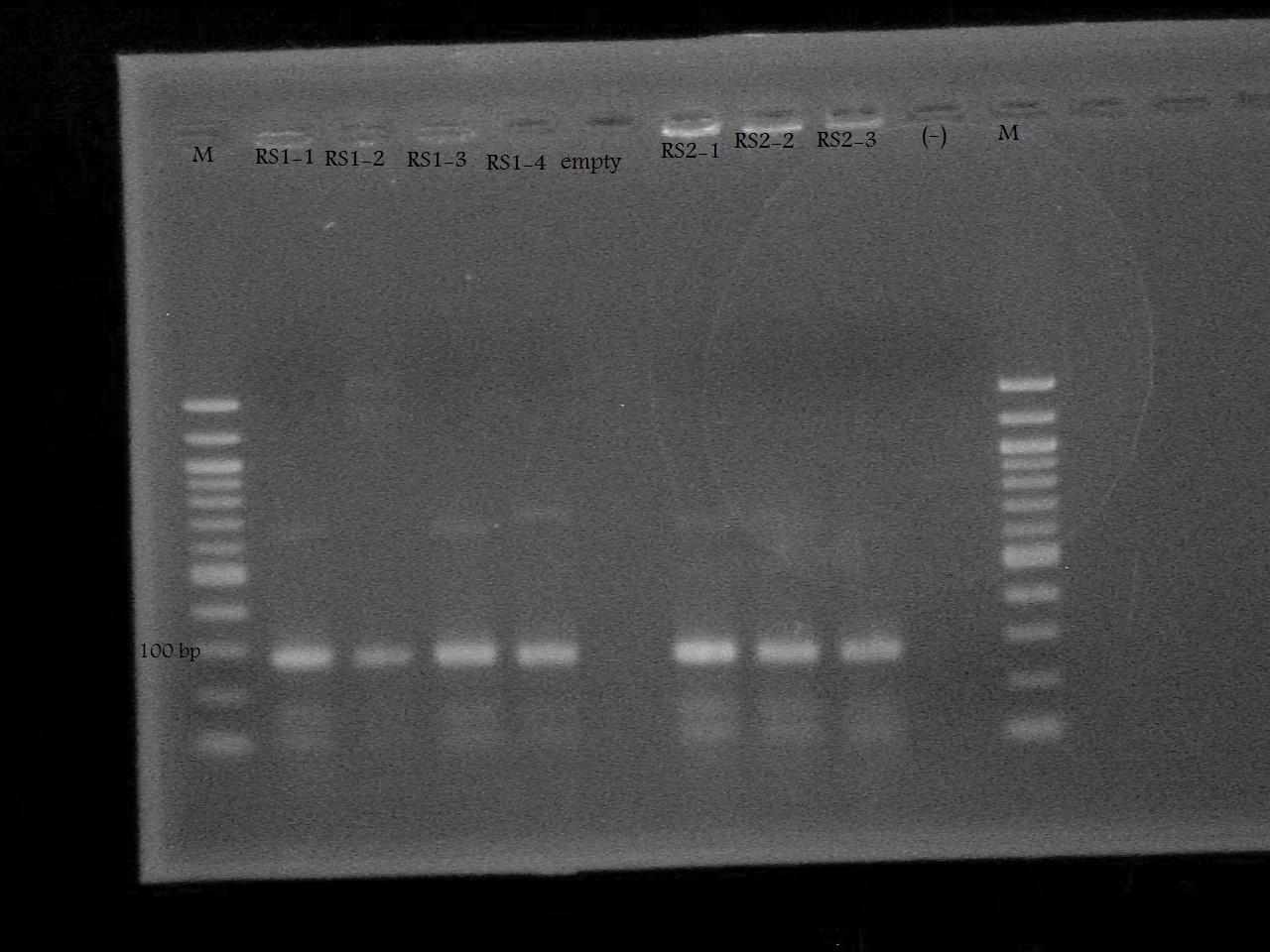
| + | -Ran the PCR Product on a 2% Agarose Gel at 100 V for 33 minutes. | ||
| + | |||
| + | [[Image:RS1 RS2 gel.jpg| 350 px]] | ||
| + | |||
| + | -Plasmid Prepped and made glycerol stocks from the RS1-1 and RS2-1 tubes of cells incubated in LB media + 100 ug/mL ampicillin. | ||
| + | |||
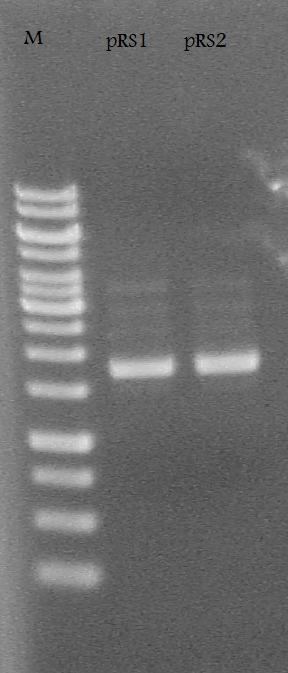
| + | Ran the pRS1 and pRS2 plasmids on a 1% Agarose Gel at 100 V for 30 minutes. | ||
| + | |||
| + | [[Image:pRS1 pRS2 gel.jpg| 150 px]] | ||
Revision as of 23:16, 3 September 2008
Back to The University of Lethbridge Main Notebook
Contents |
August 1, 2008
Nathan Puhl, Roxanne
Riboswitch 20 uL PCR. Set up 4 reactions (25 uL for each total volume). 1 uL or 1/100 pTopp and 1 uL or H1/100 PCR from July 28, 2008.
PCR conditions:
A. Initial denaturation: 98 C (3 min)
B. -Denaturation: 98 C (10 sec)
- Annealing: 55 C (30 sec)
-Extension: 72 C (15 sec)
-30 cycles
C. Final extension: 72 C (7 min)
August 2, 2008
Nathan Puhl, Roxanne
Ran riboswitch (Aug. 1, 2008) on 3% agarose gel. Results are in the hard copy lab notebook. Looks like 76 bp band will extract.
August 7, 2008
Nathan Puhl, Roxanne
Riboswitch:
Set up PCR using purified riboswitch from Aug. 5, 2008 with platinum Taq (50 uL reaction). Made Master Mix for three reactions. Master Mix:
-10x Buffer (no Mg2+): 15 uL -10 mM dNTPs: 3 uL -50 mM Mg2+: 4.5 uL -10uM RF: 3 uL -10uM RR: 3 uL -Plat. poly: 0.6 iL -H20: 120.9 uL -template: 1 uL
Cycle conditions:
A. Initial denaturation: 94 C (2 min)
B. -Denaturation: 94 C (30 sec)
- Annealing: 55 C (30 sec)
-Extension: 72 C (30 sec)
-30 cycles
C. Final extension: 72 C (7 min)
Roxanne
-Ran the PCR Product on a 3% gel using only 1uL of DNA from the riboswitch.
August 16, 2008
Nathan Puhl, Roxanne, Munima
-Restriction Digested the purified riboswitch and pSB1A7 with XbaI and SpeI, let it run for 4 hours
Nathan Puhl, Roxanne
-Ran all of the restricted pSB1A7 plasmid through a 1% agarose gel at 100V for 25 minutes. -Ran a gel extraction on the pSB1A7 cut plasmid, and ran a PCR clean-up reaction on the digested RS1 and RS2 amplicons. -Ran 1 uL of each on a 1% to quantify the amount of DNA present. -Ligated RS1 + pSB1A7, and RS2 + pSB1A7, using T4 DNA Ligase.
-1 uL of RS1/RS2 -4 uL of pSB1A7 -1 uL of 10x T4 DNA Ligase Buffer -0.33 uL of T4 DNA Ligase -3.67 uL of water
allowed the reaction to go overnight
August 17, 2008
Nathan Puhl
-Transformed DH5a cells with the pSB1A7 + RS1, and pSB1A7 + RS2 plasmids.
-Plated on semi-solid agar plates containing 100 ug/mL of ampicillin.
August 18
Christa, Nathan Puhl, Munima
-Nathan checked the plates for growth. Colonies are present.
-Ran a colony PCR of the pSB1A7 + RS1, and pSB1A7 + RS2 recombinant cells transformed by Nathan and Roxanne on August 17.
-Inoculated the cells into tubes of liquid media + 100 ug/ml of ampicillin.
Roxanne will remove the PCR tube from the thermocycler in the morning.
August 19, 2008
Roxanne
-Ran the PCR Product on a 2% Agarose Gel at 100 V for 33 minutes.
-Plasmid Prepped and made glycerol stocks from the RS1-1 and RS2-1 tubes of cells incubated in LB media + 100 ug/mL ampicillin.
Ran the pRS1 and pRS2 plasmids on a 1% Agarose Gel at 100 V for 30 minutes.
 "
"