September
From 2008.igem.org
(New page: {{Freiburg2008_Main| Content= under construction }}) |
|||
| Line 1: | Line 1: | ||
{{Freiburg2008_Main| | {{Freiburg2008_Main| | ||
Content= | Content= | ||
| - | + | <div style="font-size:18pt;"> | |
| + | <font face="Arial Rounded MT Bold" style="color:#010369">__september</font></div> | ||
| + | <br> | ||
| + | <br> | ||
| + | __NOTOC__ | ||
| + | <h3>09-10-2008</h3> | ||
| + | <br> | ||
| + | '''CMV PCR'''<br> | ||
| + | I did a PCR with primer for the CMV promotor. As DNA template I used the CMV+RLuc construct from the Ljubljana group which I isolated from the parts collection 2007.<br> | ||
| + | |||
| + | For a 50 µl reaction I used:<br> | ||
| + | 40,4 µl H2O<br> | ||
| + | 5 µl buffer (10x)<br> | ||
| + | 1,5 µl FWD Primer (15pmol)(<br> | ||
| + | 1,5 µl REV Primer (15pmol) <br> | ||
| + | 1 µl dNTP (10mM)<br> | ||
| + | 0,1 µl DNA template<br> | ||
| + | 0,5 µl Pfu Polymerase<br> | ||
| + | |||
| + | The settings for the PCR machine are the following:<br> | ||
| + | 1. T=94°C 00:02:00<br> | ||
| + | 2. T=94°C 00:00:30<br> | ||
| + | 3. T=62°C 00:00:30<br> | ||
| + | 4. T=72°C 00:01:00<br> | ||
| + | 5. GOTO 2 REP 29<br> | ||
| + | 6. T=72°C 00:10:00<br> | ||
| + | 7. HOLD 6°C<br> | ||
| + | |||
| + | I got no product. | ||
| + | |||
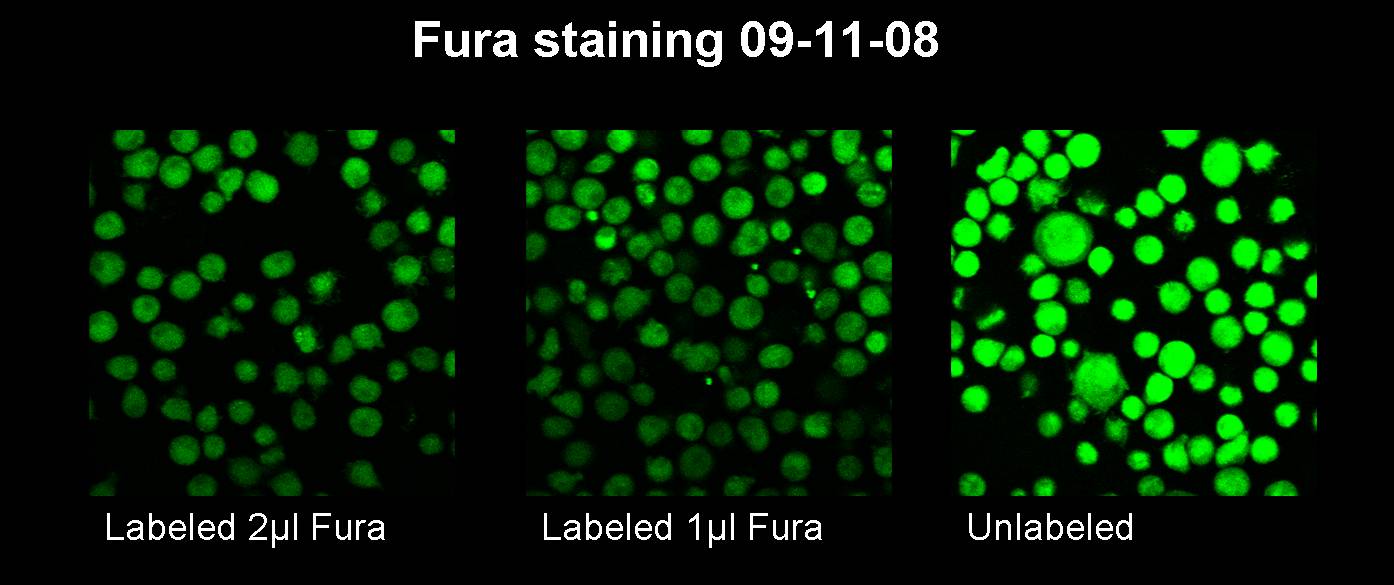
| + | <h3>09-11-2008</h3> | ||
| + | [[image:11.09.08 Test Fura staining-Freigem08.jpg |880px]]<br> | ||
| + | <br> | ||
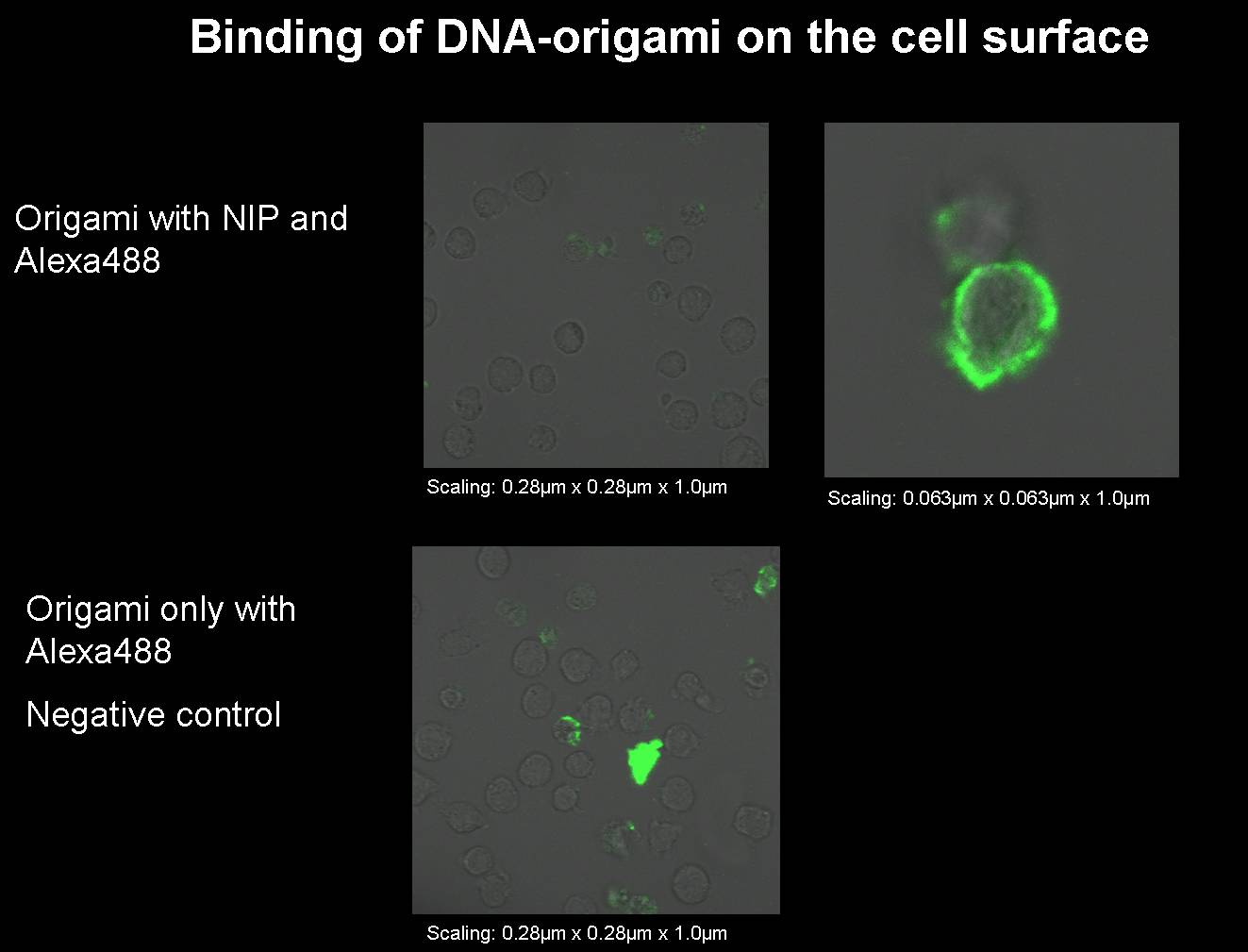
| + | [[image:11.09.08 Test Bindung Origami to alexa-Freigem08.jpg |880px]]<br> | ||
| + | |||
| + | |||
| + | <h3>09-12-2008</h3> | ||
| + | |||
| + | <html> | ||
| + | <head> | ||
| + | <meta content="text/html; charset=ISO-8859-1" | ||
| + | http-equiv="content-type"> | ||
| + | <title></title> | ||
| + | </head> | ||
| + | <body> | ||
| + | <p class="MsoNormal"><b style=""><span | ||
| + | style="" lang="EN-GB">1) Origami with NIP and | ||
| + | fluorophor for the | ||
| + | binding measurement</span><u><span style="" | ||
| + | lang="EN-GB"><o:p></o:p></span></u></b></p> | ||
| + | <p class="MsoNormal"><span style="" lang="EN-GB">We | ||
| + | had to | ||
| + | produce some new origami for our next binding measurements.<o:p></o:p></span></p> | ||
| + | <ul> | ||
| + | <li><span style="" lang="EN-GB"><span | ||
| + | style=""></span></span><span | ||
| + | style="font-family: Wingdings;" lang="EN-GB"></span><span | ||
| + | style="" lang="EN-GB">Origami with NIP and fluorophor<o:p></o:p></span></li> | ||
| + | </ul> | ||
| + | <ul> | ||
| + | <li><span style="" lang="EN-GB"><span | ||
| + | style=""></span></span><span | ||
| + | style="font-family: Wingdings;" lang="EN-GB"></span><span | ||
| + | style="" lang="EN-GB">Origami only with fluorophor | ||
| + | (without NIP); | ||
| + | negative control<o:p></o:p></span></li> | ||
| + | </ul> | ||
| + | <p class="MsoNormal"><span style="" lang="EN-GB">see | ||
| + | at the | ||
| + | protocol from </span><st1:date year="2008" day="24" | ||
| + | month="7"><span style="" lang="EN-GB">07-24-2008</span></st1:date><span | ||
| + | style="" lang="EN-GB"><o:p></o:p></span></p> | ||
| + | <p class="MsoNormal"><span style="" lang="EN-GB"><o:p></o:p></span><b | ||
| + | style=""><span style="" lang="EN-GB">2) | ||
| + | Origami for the Calciummeasurement</span><u><span | ||
| + | style="" lang="EN-GB"><o:p></o:p></span></u></b></p> | ||
| + | <ul> | ||
| + | <li><span style="" lang="EN-GB"><span | ||
| + | style=""></span></span><span | ||
| + | style="font-family: Wingdings;" lang="EN-GB"></span><span | ||
| + | style="" lang="EN-GB">Origami with NIP<o:p></o:p></span></li> | ||
| + | </ul> | ||
| + | <ul> | ||
| + | <li><span style="" lang="EN-GB"><span | ||
| + | style=""></span></span><span | ||
| + | style="font-family: Wingdings;" lang="EN-GB"></span><span | ||
| + | style="" lang="EN-GB">Origami without NIP (negative | ||
| + | control)<o:p></o:p></span></li> | ||
| + | </ul> | ||
| + | <p class="MsoNormal" | ||
| + | style="margin-left: 35.4pt; text-indent: -35.4pt;"><span | ||
| + | style="" lang="EN-GB">see at the | ||
| + | protocol from </span><st1:date year="2008" day="24" | ||
| + | month="7"><span style="" lang="EN-GB">07-24-2008</span></st1:date><span | ||
| + | style="" lang="EN-GB"><o:p></o:p></span></p> | ||
| + | <p class="MsoNormal" | ||
| + | style="margin-left: 35.4pt; text-indent: -35.4pt;"><span | ||
| + | style="" lang="EN-GB">To increase the concentration | ||
| + | of | ||
| + | origami we also made to probes with the double amount<o:p></o:p></span></p> | ||
| + | <p class="MsoNormal" | ||
| + | style="margin-left: 35.4pt; text-indent: -35.4pt;"><span | ||
| + | style="" lang="EN-GB">ingredients of the protocol | ||
| + | from </span><st1:date year="2008" day="24" | ||
| + | month="7"><span style="" lang="EN-GB">07-24-2008</span></st1:date><span | ||
| + | style="" lang="EN-GB"><o:p></o:p></span></p> | ||
| + | <table class="MsoTableGrid" | ||
| + | style="border: medium none ; border-collapse: collapse;" | ||
| + | border="1" cellpadding="0" cellspacing="0"> | ||
| + | <tbody> | ||
| + | <tr style=""> | ||
| + | <td | ||
| + | style="border: 1pt solid windowtext; padding: 0cm 5.4pt; width: 107.4pt;" | ||
| + | valign="top" width="143"> | ||
| + | <p class="MsoNormal"><span style="" | ||
| + | lang="EN-GB"><o:p> </o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: solid solid solid none; border-color: windowtext windowtext windowtext -moz-use-text-color; border-width: 1pt 1pt 1pt medium; padding: 0cm 5.4pt; width: 90pt;" | ||
| + | valign="top" width="120"> | ||
| + | <p class="MsoNormal"><span style="" | ||
| + | lang="EN-GB">Origami with NIP (6x (1:5)) [µl]<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: solid solid solid none; border-color: windowtext windowtext windowtext -moz-use-text-color; border-width: 1pt 1pt 1pt medium; padding: 0cm 5.4pt; width: 108pt;" | ||
| + | valign="top" width="144"> | ||
| + | <p class="MsoNormal"><span style="" | ||
| + | lang="EN-GB">Origami without NIP (6x (1:5)) [µl]<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr style=""> | ||
| + | <td | ||
| + | style="border-style: none solid solid; border-color: -moz-use-text-color windowtext windowtext; border-width: medium 1pt 1pt; padding: 0cm 5.4pt; width: 107.4pt;" | ||
| + | valign="top" width="143"> | ||
| + | <p class="MsoNormal"><span style="" | ||
| + | lang="EN-GB">Oligos-Pool<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: none solid solid none; border-color: -moz-use-text-color windowtext windowtext -moz-use-text-color; border-width: medium 1pt 1pt medium; padding: 0cm 5.4pt; width: 90pt;" | ||
| + | valign="top" width="120"> | ||
| + | <p class="MsoNormal" style="text-align: right;" | ||
| + | align="right"><span style="" lang="EN-GB">43,68<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: none solid solid none; border-color: -moz-use-text-color windowtext windowtext -moz-use-text-color; border-width: medium 1pt 1pt medium; padding: 0cm 5.4pt; width: 108pt;" | ||
| + | valign="top" width="144"> | ||
| + | <p class="MsoNormal" style="text-align: right;" | ||
| + | align="right"><span style="" lang="EN-GB">43,68<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr style=""> | ||
| + | <td | ||
| + | style="border-style: none solid solid; border-color: -moz-use-text-color windowtext windowtext; border-width: medium 1pt 1pt; padding: 0cm 5.4pt; width: 107.4pt;" | ||
| + | valign="top" width="143"> | ||
| + | <p class="MsoNormal"><span style="" | ||
| + | lang="EN-GB">remainders<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: none solid solid none; border-color: -moz-use-text-color windowtext windowtext -moz-use-text-color; border-width: medium 1pt 1pt medium; padding: 0cm 5.4pt; width: 90pt;" | ||
| + | valign="top" width="120"> | ||
| + | <p class="MsoNormal" style="text-align: right;" | ||
| + | align="right"><span style="" lang="EN-GB">2,4<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: none solid solid none; border-color: -moz-use-text-color windowtext windowtext -moz-use-text-color; border-width: medium 1pt 1pt medium; padding: 0cm 5.4pt; width: 108pt;" | ||
| + | valign="top" width="144"> | ||
| + | <p class="MsoNormal" style="text-align: right;" | ||
| + | align="right"><span style="" lang="EN-GB">2,4<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr style=""> | ||
| + | <td | ||
| + | style="border-style: none solid solid; border-color: -moz-use-text-color windowtext windowtext; border-width: medium 1pt 1pt; padding: 0cm 5.4pt; width: 107.4pt;" | ||
| + | valign="top" width="143"> | ||
| + | <p class="MsoNormal"><span style="" | ||
| + | lang="EN-GB">MgAc<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: none solid solid none; border-color: -moz-use-text-color windowtext windowtext -moz-use-text-color; border-width: medium 1pt 1pt medium; padding: 0cm 5.4pt; width: 90pt;" | ||
| + | valign="top" width="120"> | ||
| + | <p class="MsoNormal" style="text-align: right;" | ||
| + | align="right"><span style="" lang="EN-GB">1<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: none solid solid none; border-color: -moz-use-text-color windowtext windowtext -moz-use-text-color; border-width: medium 1pt 1pt medium; padding: 0cm 5.4pt; width: 108pt;" | ||
| + | valign="top" width="144"> | ||
| + | <p class="MsoNormal" style="text-align: right;" | ||
| + | align="right"><span style="" lang="EN-GB">1<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr style=""> | ||
| + | <td | ||
| + | style="border-style: none solid solid; border-color: -moz-use-text-color windowtext windowtext; border-width: medium 1pt 1pt; padding: 0cm 5.4pt; width: 107.4pt;" | ||
| + | valign="top" width="143"> | ||
| + | <p class="MsoNormal"><span style="" | ||
| + | lang="EN-GB">Phage DNA (448,4 mg/µl)<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: none solid solid none; border-color: -moz-use-text-color windowtext windowtext -moz-use-text-color; border-width: medium 1pt 1pt medium; padding: 0cm 5.4pt; width: 90pt;" | ||
| + | valign="top" width="120"> | ||
| + | <p class="MsoNormal" style="text-align: right;" | ||
| + | align="right"><span style="" lang="EN-GB">33.6<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: none solid solid none; border-color: -moz-use-text-color windowtext windowtext -moz-use-text-color; border-width: medium 1pt 1pt medium; padding: 0cm 5.4pt; width: 108pt;" | ||
| + | valign="top" width="144"> | ||
| + | <p class="MsoNormal" style="text-align: right;" | ||
| + | align="right"><span style="" lang="EN-GB">33.6<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr style=""> | ||
| + | <td | ||
| + | style="border-style: none solid solid; border-color: -moz-use-text-color windowtext windowtext; border-width: medium 1pt 1pt; padding: 0cm 5.4pt; width: 107.4pt;" | ||
| + | valign="top" width="143"> | ||
| + | <p class="MsoNormal"><span style="" | ||
| + | lang="EN-GB">NIP-Oligo<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: none solid solid none; border-color: -moz-use-text-color windowtext windowtext -moz-use-text-color; border-width: medium 1pt 1pt medium; padding: 0cm 5.4pt; width: 90pt;" | ||
| + | valign="top" width="120"> | ||
| + | <p class="MsoNormal" style="text-align: right;" | ||
| + | align="right"><span style="" lang="EN-GB">1,68<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: none solid solid none; border-color: -moz-use-text-color windowtext windowtext -moz-use-text-color; border-width: medium 1pt 1pt medium; padding: 0cm 5.4pt; width: 108pt;" | ||
| + | valign="top" width="144"> | ||
| + | <p class="MsoNormal"><span style="" | ||
| + | lang="EN-GB">---------------------<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr style=""> | ||
| + | <td | ||
| + | style="border-style: none solid solid; border-color: -moz-use-text-color windowtext windowtext; border-width: medium 1pt 1pt; padding: 0cm 5.4pt; width: 107.4pt;" | ||
| + | valign="top" width="143"> | ||
| + | <p class="MsoNormal"><span style="" | ||
| + | lang="EN-GB">Pool oligo without fluorophor<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: none solid solid none; border-color: -moz-use-text-color windowtext windowtext -moz-use-text-color; border-width: medium 1pt 1pt medium; padding: 0cm 5.4pt; width: 90pt;" | ||
| + | valign="top" width="120"> | ||
| + | <p class="MsoNormal" style="text-align: right;" | ||
| + | align="right"><span style="" lang="EN-GB">0,72<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: none solid solid none; border-color: -moz-use-text-color windowtext windowtext -moz-use-text-color; border-width: medium 1pt 1pt medium; padding: 0cm 5.4pt; width: 108pt;" | ||
| + | valign="top" width="144"> | ||
| + | <p class="MsoNormal" style="text-align: right;" | ||
| + | align="right"><span style="" lang="EN-GB">0,72<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | </tr> | ||
| + | <tr style=""> | ||
| + | <td | ||
| + | style="border-style: none solid solid; border-color: -moz-use-text-color windowtext windowtext; border-width: medium 1pt 1pt; padding: 0cm 5.4pt; width: 107.4pt;" | ||
| + | valign="top" width="143"> | ||
| + | <p class="MsoNormal"><span style="" | ||
| + | lang="EN-GB">Oligo without NIP<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: none solid solid none; border-color: -moz-use-text-color windowtext windowtext -moz-use-text-color; border-width: medium 1pt 1pt medium; padding: 0cm 5.4pt; width: 90pt;" | ||
| + | valign="top" width="120"> | ||
| + | <p class="MsoNormal" style="text-align: right;" | ||
| + | align="right"><span style="" lang="EN-GB">-------------------<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | <td | ||
| + | style="border-style: none solid solid none; border-color: -moz-use-text-color windowtext windowtext -moz-use-text-color; border-width: medium 1pt 1pt medium; padding: 0cm 5.4pt; width: 108pt;" | ||
| + | valign="top" width="144"> | ||
| + | <p class="MsoNormal" style="text-align: right;" | ||
| + | align="right"><span style="" lang="EN-GB">1,68<o:p></o:p></span></p> | ||
| + | </td> | ||
| + | </tr> | ||
| + | </tbody> | ||
| + | </table> | ||
| + | <p class="MsoNormal" | ||
| + | style="margin-left: 35.4pt; text-indent: -35.4pt;"><b | ||
| + | style=""><span style="" lang="EN-GB"><o:p></o:p></span><span | ||
| + | style="" lang="EN-GB">3) Master cycler</span><u><span | ||
| + | style="" lang="EN-GB"><o:p></o:p></span></u></b></p> | ||
| + | <p class="MsoNormal" | ||
| + | style="margin-left: 35.4pt; text-indent: -35.4pt;"><span | ||
| + | style="" lang="EN-GB">The origamis were produced in | ||
| + | the | ||
| + | mastercycler as explained before.<o:p></o:p></span></p> | ||
| + | <p class="MsoNormal" | ||
| + | style="margin-left: 35.4pt; text-indent: -35.4pt;"><b | ||
| + | style=""><o:p> </o:p><span style="" | ||
| + | lang="EN-GB">4) Purification of the DNA Origami</span><u><span | ||
| + | style="" lang="EN-GB"><o:p></o:p></span></u></b></p> | ||
| + | <p class="MsoNormal" | ||
| + | style="margin-left: 35.4pt; text-indent: -35.4pt;"><span | ||
| + | style="" lang="EN-GB">Was done as before<o:p></o:p></span></p> | ||
| + | </body> | ||
| + | </html> | ||
| + | |||
| + | '''5) Digestion of CMV+Rluc'''<br> | ||
| + | Digestion with EcoRV und FspI(3h at 37°C) | ||
| + | <ul> | ||
| + | <li> 5µl plasmid | ||
| + | <li> 10µl H2O | ||
| + | <li> 0,5µl enzyme | ||
| + | <li> 2,5µl buffer (2) | ||
| + | <li> 0,5µl BSA | ||
| + | </ul> | ||
| + | [[image:Verdau_CMVRluc_EcoRV_FspI_klein.jpg]]<br> | ||
| + | |||
| + | '''6) CMV PCR'''<br> | ||
| + | I tried the same PCR with different annealing temperatures. The optimal annealing temperature is 62°C. I used a gradient between 58°C and 62°C. I again got no products. | ||
| + | |||
| + | <h3>09-15-2008</h3> | ||
| + | '''CMV PCR'''<br> | ||
| + | I again tried the CMV PCR, but this time with different polymerases. I used the Taq Polymerase and a Mix. I also tried one approach with the complete plasmid and one with the digested plasmid (see digestion of CMV+Rluc) | ||
| + | I got products in the approach with taq polymerase as well as in the approach with the Mix. I cut out 3 bands from each approach. | ||
| + | |||
| + | <h3>09-16-2008</h3> | ||
| + | '''Gel purification'''<br> | ||
| + | I did a gel purification of the PCR products. | ||
| + | |||
| + | '''Digestion of the PCR products and the transfectionvector'''<br> | ||
| + | Digestion with EcoRI and PstI. Then Kathrin did a ligation. | ||
| + | |||
| + | <h3>09-18-2008</h3> | ||
| + | '''Transformation'''<br> | ||
| + | I did a transformation with the ligation product. | ||
| + | |||
| + | <h3>09-19-2008</h3> | ||
| + | '''Transformation'''<br> | ||
| + | There were no colonies on the plates. | ||
| + | |||
| + | <h3>09-20-2008</h3> | ||
| + | '''Digestion of the PCR products and the transfectionvector'''<br> | ||
| + | Now I used the restrictionenzymes XbaI and SpeI. | ||
| + | |||
| + | '''Gel purification and ligation'''<br> | ||
| + | The digested PCR products and the vector. | ||
| + | |||
| + | <h3>09-21-2008</h3> | ||
| + | '''Transformation of the ligation'''<br> | ||
| + | I used 10 µl of the ligation and used RV 308 cells | ||
}} | }} | ||
Revision as of 18:32, 9 October 2008
|
__september
09-10-2008
For a 50 µl reaction I used: The settings for the PCR machine are the following: I got no product. 09-11-2008
09-12-2008
1) Origami with NIP and
fluorophor for the
binding measurement We
had to
produce some new origami for our next binding measurements.
see
at the
protocol from
see at the
protocol from To increase the concentration
of
origami we also made to probes with the double amount ingredients of the protocol
from
The origamis were produced in
the
mastercycler as explained before. Was done as before 5) Digestion of CMV+Rluc
File:Verdau CMVRluc EcoRV FspI klein.jpg 6) CMV PCR 09-15-2008CMV PCR 09-16-2008Gel purification Digestion of the PCR products and the transfectionvector 09-18-2008Transformation 09-19-2008Transformation 09-20-2008Digestion of the PCR products and the transfectionvector Gel purification and ligation 09-21-2008Transformation of the ligation |
 "
"