Team:Chiba/jk/γ/Trl
From 2008.igem.org
(Difference between revisions)
(→Protocol) |
(→purpose) |
||
| (6 intermediate revisions not shown) | |||
| Line 5: | Line 5: | ||
==Time Responce Liquid== | ==Time Responce Liquid== | ||
===purpose=== | ===purpose=== | ||
| - | |||
<br>To find the earliest output gene after induction | <br>To find the earliest output gene after induction | ||
| - | === | + | ===Reporter=== |
| - | + | *Fluorescent Protein | |
| - | * | + | :*GFP |
| - | * | + | ::*pGFPuv |
| - | * | + | ::BBa_T9002 |
| - | + | :*Venus YFP | |
| - | * | + | ::*BBa_K084003 |
| - | * | + | :::*pLac-Venus YFP |
| - | *X-gal | + | :*mCherry |
| - | * | + | pLac-mCherry |
| - | + | *β-gal (X-gal assay) | |
| + | :*pUC19(plac-LacZα) | ||
| - | === | + | ===Equipment=== |
| - | + | :shaking incubator | |
| - | : | + | ::Innova 4200 Benchtop or Floor-Stackable Incubator Shaker(37°C) |
| - | : | + | :46-well plate(deep well) |
| - | : | + | :96-well plate(deep well) |
| - | : | + | :Fluoroskan Ascent 2.5(program:Ascent Software Version 2.6) |
| - | + | :Beckman Allegratm X-12R Centrifuga(Beckman Coulter) | |
| - | + | ||
| - | : | + | |
| - | : | + | |
| - | + | ||
| - | + | ||
| - | + | ||
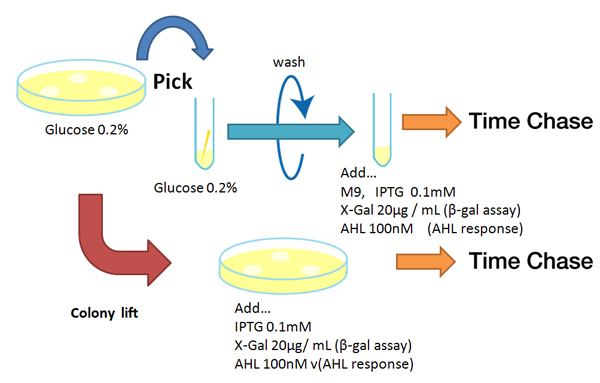
| - | + | ===Method=== | |
| - | : | + | [[Image:Reporter method Chiba.jpg|rifht|thumb|'''Fig. '''レポーターのタイムレスポンスの実験方法]] |
| - | : | + | Strain:XL10G Kan<sup>R</sup> |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | *Liquid medium experiment | |
| - | + | #Pre-culture | |
| - | + | ##Picked and cultured the following plate in 2mL of LB: | |
| - | + | ###LB-Amp+0.2 % Glucose, (pGFPuv, pLac-Venus YFP, pLac-mCherry, pUC19) | |
| - | + | ###LB-Amp, ([http://partsregistry.org/Part:BBa_T9002 BBa_T9002], [http://partsregistry.org/Part:BBa_K084003 BBa_K084003]) | |
| - | + | ##Cultured at 37°C for 12h. | |
| - | + | #Culture | |
| + | ##Dilute pre-cultures and add to new LB medium. | ||
| + | ###LB-Amp+0.2 % Glucose, (pGFPuv, pLac-Venus YFP, pLac-mCherry, pUC19) | ||
| + | ###LB-Amp, ([http://partsregistry.org/Part:BBa_T9002 BBa_T9002], [http://partsregistry.org/Part:BBa_K084003 BBa_K084003]) | ||
| + | ##Cultured at 37°C for about 6 h | ||
| + | #Wash | ||
| + | ##Transfer 10mL each of the culture to 50mL centrifuge tubes. | ||
| + | ##Centrifuged for 6min at 3600rpm,20°C and discarded the supernatant. | ||
| + | ##Add physiological saline and resuspention | ||
| + | ##Repeated wash twice. | ||
| + | ##Add M9 minimal medium. | ||
| + | #Mix | ||
| + | ##Dispens each culture into 48-well deep well or 96-well deep well | ||
| + | ##Add IPTG fainal conc. 0.2 mM (pGFPuv, pLac-Venus YFP, pLac-mCherry, pUC19) | ||
| + | ##Add AHL fainal conc. 100nM ([http://partsregistry.org/Part:BBa_T9002 BBa_T9002], [http://partsregistry.org/Part:BBa_K084003 BBa_K084003]) | ||
| + | #Measure fluorescence intensity every 1 h. | ||
| - | + | ===Result=== | |
*[[TEam:Chiba/jk/γ/Trl/0828|28,August,2008]] | *[[TEam:Chiba/jk/γ/Trl/0828|28,August,2008]] | ||
*[[TEam:Chiba/jk/γ/Trl/0905|5,September,2008]] | *[[TEam:Chiba/jk/γ/Trl/0905|5,September,2008]] | ||
*[[TEam:Chiba/jk/γ/Trl/0910|10,September,2008]] | *[[TEam:Chiba/jk/γ/Trl/0910|10,September,2008]] | ||
*[[TEam:Chiba/jk/γ/Trl/0914|14,September,2008]] | *[[TEam:Chiba/jk/γ/Trl/0914|14,September,2008]] | ||
| + | |||
| + | '''Discussion''' | ||
| + | Discussion | ||
| + | |||
| + | |||
| + | The purpose of this project is to alter the time required for expression. | ||
| + | |||
| + | * By increasing the concentration of X-gal, the time required for | ||
| + | expression to be observable decreased, but this still takes longer | ||
| + | than the time required when using fluorescent proteins. | ||
| + | |||
| + | |||
| + | *Using β-gal, which uses X-gal as substrate, the time required for | ||
| + | output may increase because the substrate concentration decreases with | ||
| + | time. | ||
| + | |||
| + | For the above reasons, fluorescence proteins are more suited for our purposes. | ||
| + | |||
| + | Of the fluorescent proteins, GFP and YFP fluorescence was observable | ||
| + | at earlier stages. | ||
| + | |||
| + | |||
| + | * But to the naked eye, GFP could be observed more easily. | ||
| + | |||
| + | Therefore, we chose GFP as our output reporter. | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
{| style="color:white;background-color:Maroon" cellpadding="3" cellspacing="3" border="1" bordercolor="white" width="100%" align="center" | {| style="color:white;background-color:Maroon" cellpadding="3" cellspacing="3" border="1" bordercolor="white" width="100%" align="center" | ||
Latest revision as of 04:41, 30 October 2008
Contents |
Time Responce Liquid
purpose
To find the earliest output gene after induction
Reporter
- Fluorescent Protein
- GFP
- pGFPuv
- BBa_T9002
- Venus YFP
- BBa_K084003
- pLac-Venus YFP
- mCherry
pLac-mCherry
- β-gal (X-gal assay)
- pUC19(plac-LacZα)
Equipment
- shaking incubator
- Innova 4200 Benchtop or Floor-Stackable Incubator Shaker(37°C)
- 46-well plate(deep well)
- 96-well plate(deep well)
- Fluoroskan Ascent 2.5(program:Ascent Software Version 2.6)
- Beckman Allegratm X-12R Centrifuga(Beckman Coulter)
Method
Strain:XL10G KanR
- Liquid medium experiment
- Pre-culture
- Picked and cultured the following plate in 2mL of LB:
- LB-Amp+0.2 % Glucose, (pGFPuv, pLac-Venus YFP, pLac-mCherry, pUC19)
- LB-Amp, ([http://partsregistry.org/Part:BBa_T9002 BBa_T9002], [http://partsregistry.org/Part:BBa_K084003 BBa_K084003])
- Cultured at 37°C for 12h.
- Picked and cultured the following plate in 2mL of LB:
- Culture
- Dilute pre-cultures and add to new LB medium.
- LB-Amp+0.2 % Glucose, (pGFPuv, pLac-Venus YFP, pLac-mCherry, pUC19)
- LB-Amp, ([http://partsregistry.org/Part:BBa_T9002 BBa_T9002], [http://partsregistry.org/Part:BBa_K084003 BBa_K084003])
- Cultured at 37°C for about 6 h
- Dilute pre-cultures and add to new LB medium.
- Wash
- Transfer 10mL each of the culture to 50mL centrifuge tubes.
- Centrifuged for 6min at 3600rpm,20°C and discarded the supernatant.
- Add physiological saline and resuspention
- Repeated wash twice.
- Add M9 minimal medium.
- Mix
- Dispens each culture into 48-well deep well or 96-well deep well
- Add IPTG fainal conc. 0.2 mM (pGFPuv, pLac-Venus YFP, pLac-mCherry, pUC19)
- Add AHL fainal conc. 100nM ([http://partsregistry.org/Part:BBa_T9002 BBa_T9002], [http://partsregistry.org/Part:BBa_K084003 BBa_K084003])
- Measure fluorescence intensity every 1 h.
Result
Discussion Discussion
The purpose of this project is to alter the time required for expression.
- By increasing the concentration of X-gal, the time required for
expression to be observable decreased, but this still takes longer than the time required when using fluorescent proteins.
- Using β-gal, which uses X-gal as substrate, the time required for
output may increase because the substrate concentration decreases with time.
For the above reasons, fluorescence proteins are more suited for our purposes.
Of the fluorescent proteins, GFP and YFP fluorescence was observable at earlier stages.
- But to the naked eye, GFP could be observed more easily.
Therefore, we chose GFP as our output reporter.
| ホーム | メンバー紹介 | プロジェクト紹介 | Parts Submitted to the Registry | モデリング | ノート |
|---|
 "
"