Team:Imperial College/Summary
From 2008.igem.org
m |
m |
||
| Line 42: | Line 42: | ||
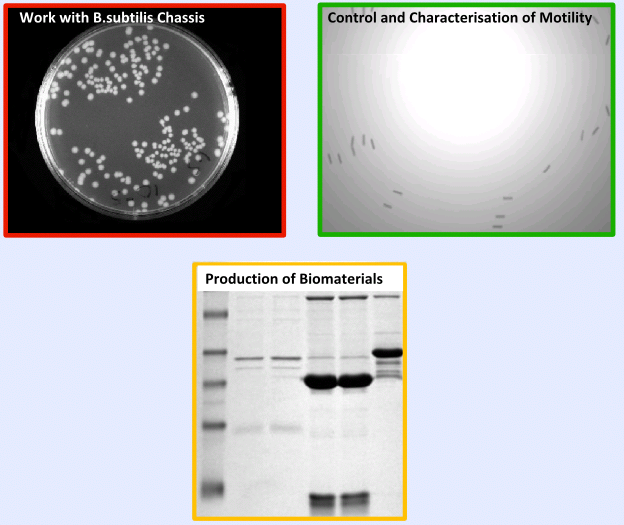
*'''Production of Biomaterials in ''B. subtilis''''' | *'''Production of Biomaterials in ''B. subtilis''''' | ||
| - | If you'd like to see more information on the key results from the testing and validation, you can find it on the [https://2008.igem.org/Team:Imperial_College/Major_Results Results Page]. | + | If you'd like to see more information on the key results from the testing and validation, you can find it on the [https://2008.igem.org/Team:Imperial_College/Major_Results '''Results Page''']. |
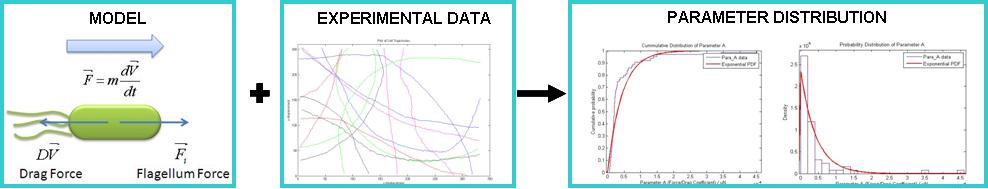
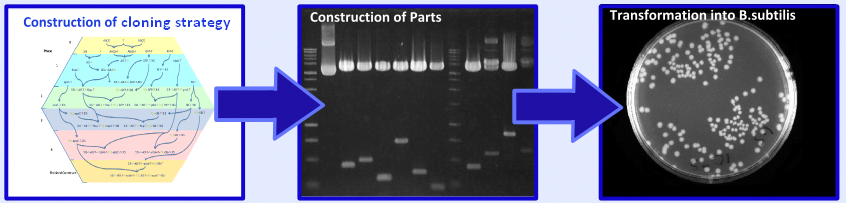
=====Results===== | =====Results===== | ||
|[[Image:Result.PNG|center|300px]]}} | |[[Image:Result.PNG|center|300px]]}} | ||
Revision as of 02:39, 30 October 2008
Project Summary
|
|||||||||||||||||||||||||||
 "
"