Team:Caltech/Project
From 2008.igem.org
Caltechigem (Talk | contribs) |
Caltechigem (Talk | contribs) |
||
| Line 42: | Line 42: | ||
{{!}} | {{!}} | ||
Folate, a term which encompasses the various forms of the vitamin B9, is an essential vitamin involved in everyday cell functions such as DNA replication. Unable to naturally produce folate, humans must obtain it from vegetables or folate-supplements. In regions with little or no access to these foods, folate deficiencies can cause serious birth defects. One possible solution to alleviate the effects of folate deficiency is to engineer a strain of gut microbes to produce bioavailable folate directly in the colon. A total of four heterologous genes, two from the folate biosynthesis gene cluster and two from the paraaminobenzoic acid (pABA) synthesis pathway, were tested. Using standardized genetic sequences, folate biosynthesis genes extracted from the ''Lactoccocus lactis'' genome were cloned into Biobricks plasmids, transformed into ''Escherichia coli'' and overexpressed. The effects of overexpression | Folate, a term which encompasses the various forms of the vitamin B9, is an essential vitamin involved in everyday cell functions such as DNA replication. Unable to naturally produce folate, humans must obtain it from vegetables or folate-supplements. In regions with little or no access to these foods, folate deficiencies can cause serious birth defects. One possible solution to alleviate the effects of folate deficiency is to engineer a strain of gut microbes to produce bioavailable folate directly in the colon. A total of four heterologous genes, two from the folate biosynthesis gene cluster and two from the paraaminobenzoic acid (pABA) synthesis pathway, were tested. Using standardized genetic sequences, folate biosynthesis genes extracted from the ''Lactoccocus lactis'' genome were cloned into Biobricks plasmids, transformed into ''Escherichia coli'' and overexpressed. The effects of overexpression | ||
| - | were measured in terms of total folate and paraaminobenzoic acid levels. PABA, an intermediate in folate synthesis, was detected using [[Team:Caltech/Protocols/PABA_HPLC_assay|<font style="color:#BB4400">high performance liquid chromatography</font>]] (HPLC). Folate detection was achieved via a [[Team:Caltech/Protocols/Folate_assay|<font style="color:#BB4400">microbiological assay</font>]]. A measurable increase in folate production in ''E. coli'' | + | were measured in terms of total folate and paraaminobenzoic acid levels. PABA, an intermediate in folate synthesis, was detected using [[Team:Caltech/Protocols/PABA_HPLC_assay|<font style="color:#BB4400">high performance liquid chromatography</font>]] (HPLC). Folate detection was achieved via a [[Team:Caltech/Protocols/Folate_assay|<font style="color:#BB4400">microbiological assay</font>]]. A measurable increase in folate production in ''E. coli'' provides proof-of-concept for both the feasibility of engineering overproduction of folate in ''E. coli'', as well as using standardized genetic components to do so. |
{{!}}{{navimg|xsize=193|ysize=288|image=Folate_foods.jpg|link=Team:Caltech/Project/Vitamins}} | {{!}}{{navimg|xsize=193|ysize=288|image=Folate_foods.jpg|link=Team:Caltech/Project/Vitamins}} | ||
{{!}}} | {{!}}} | ||
| Line 51: | Line 51: | ||
{{!}}{{navimg|xsize=220|ysize=231|image=Differentiation.jpg|link=Team:Caltech/Project/Population_Variation}} | {{!}}{{navimg|xsize=220|ysize=231|image=Differentiation.jpg|link=Team:Caltech/Project/Population_Variation}} | ||
{{!}} | {{!}} | ||
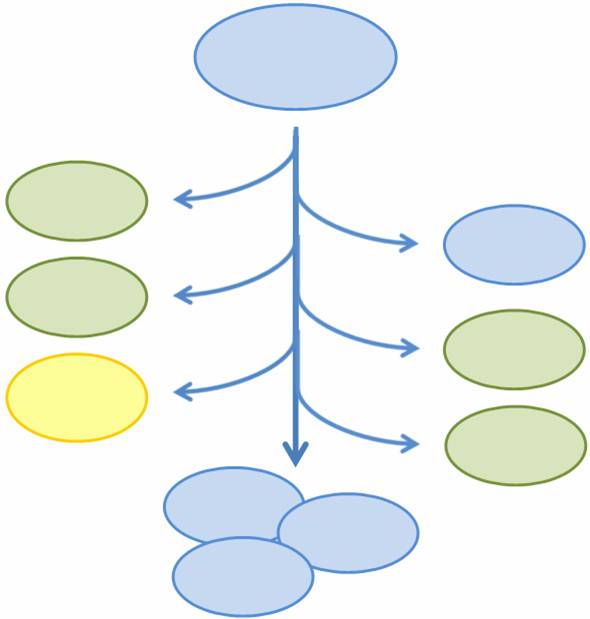
| - | As more complicated and interconnected biological circuits are | + | As more complicated and interconnected biological circuits are built, there is an increasing need for the simple integration of multiple functions into a single bacterial cell line. However, some of these functions may be incompatible or may kill the cell, such that each cell can only express a single function at any time or must be regenerated if the functionality is fatal. We aim to combine multiple mutually exclusive and potentially fatal functions into a single bacterial cell line that, as a population, exhibits the entire set of functions. |
| - | In particular, we want only one of the four | + | In particular, we want only one of the four sub-projects described above to be turned on in any given cell, or else the cell may be overburdened by our constructs. At the same time, we want our bacterial population to have the capability to exhibit all four functions. In addition, three of the four sub-projects result in the death of the host cell through different methods of self-induced lysis. Therefore, we need a system that is able to combine all sub-projects into one coherent system and that allows for self-renewal of the population. |
| - | We propose a system in which bacterial cells | + | We propose a system in which a single bacterial strain encodes all four functions in its genome and can access each function independently through probabalistic molecular events. The engineered bacterial cells start in an undifferentiated state and randomly differentiate into one of three possible final states. To build this system, we designed two genetic devices. One relies on DNA polymerase slippage upon replication of a long stretch of short nucleotide repeats, a phenomenon termed slipped-strand mispairing (SSM). The other device uses the recombinase protein FimE to flip DNA segments. The two devices can form a system that permits the novel introduction of random multi-state differentiation into bacterial cells. Our experimental results demonstrate that short nucleotide repeats can be used as a stochastic switch and that FimE activity depends on the length of the segment being flipped. |
{{!}}} | {{!}}} | ||
}} | }} | ||
Revision as of 06:57, 25 October 2008
|
People
|
Subprojects
Note: Click on the subproject title or picture for a detailed description of the subproject
Oxidative Burst
Phage Pathogen Defense
Lactose Intolerance
Vitamin Production
Population Variation
|
 "
"