Team:Caltech/Protocols/rcsA Lysogen Induction
From 2008.igem.org
(Difference between revisions)
| Line 8: | Line 8: | ||
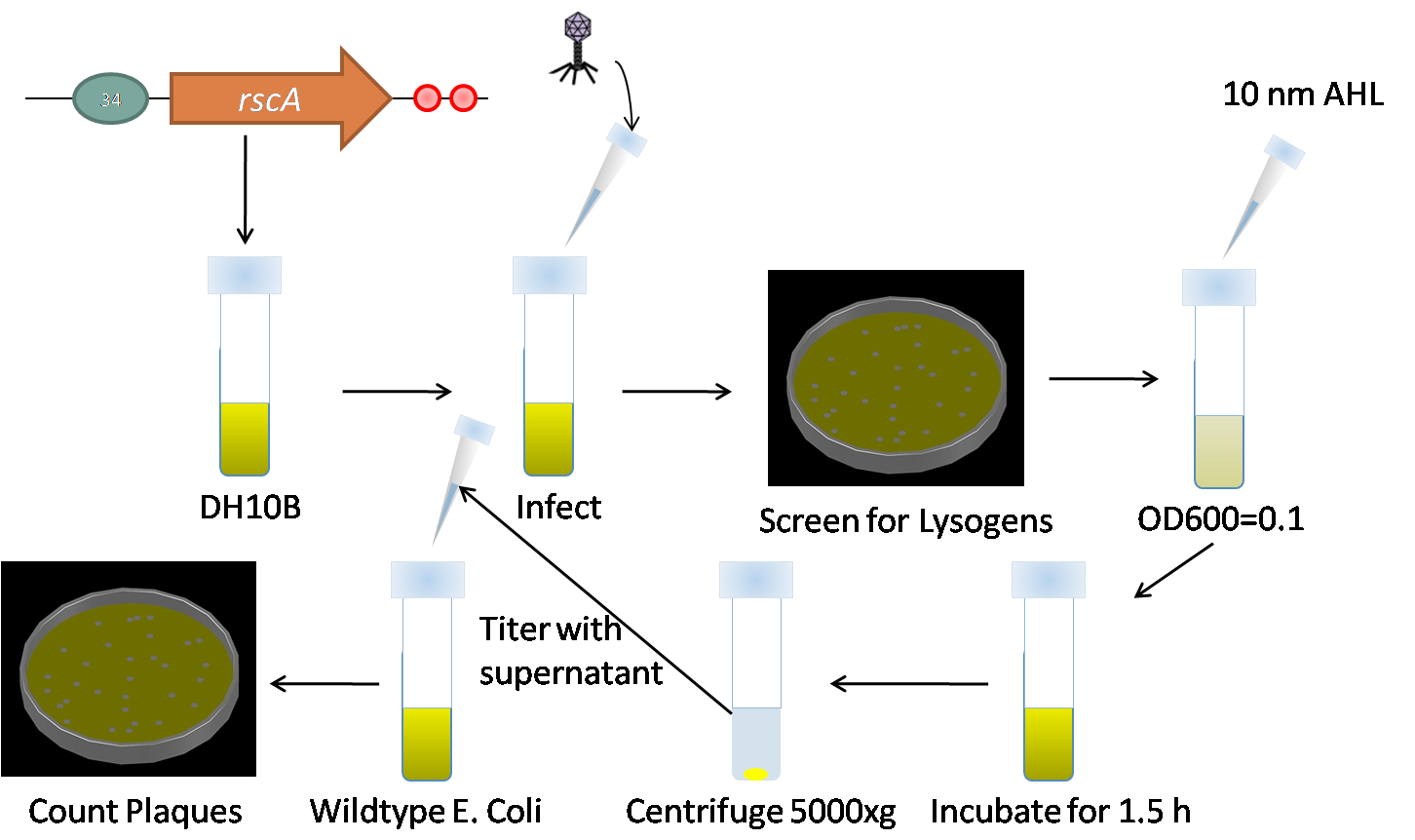
#Grow fresh overnight cultures of lysogens of bacteria carrying the rcsA construct. | #Grow fresh overnight cultures of lysogens of bacteria carrying the rcsA construct. | ||
| - | #Grow fresh overnight cultures wildtype E. coli. | + | #Grow fresh overnight cultures wildtype E. coli. [[Image:Induction.png|right|thumb|400px|Diagram of the induction process.]] |
| - | [[Image:Induction.png|right|thumb|400px|Diagram of the induction process.]] | + | |
#Make a 1:1000 dilution of lysogen culture and incubate the cultures until they reach an OD600 of 0.1 | #Make a 1:1000 dilution of lysogen culture and incubate the cultures until they reach an OD600 of 0.1 | ||
#* More specifically, when the culture is swirled, cloudiness is observed. Check OD on plate reader as it is important to keep OD constant between trials and experiments. | #* More specifically, when the culture is swirled, cloudiness is observed. Check OD on plate reader as it is important to keep OD constant between trials and experiments. | ||
Latest revision as of 05:17, 24 October 2008
|
People
|
rcsA Lysogen Induction
|
 "
"