Team:Harvard/Dailybook/Week8/Widgetry
From 2008.igem.org
Contents |
Goals for this week
- Run co-culture test with DH5α, DH5α/pUC19, and Shewie
- Move chambers to the "dark" to prevent a reaction from happening with light and oxygen
- Test light-repressible system on plates
Monday: August 11, 2008
- cleaned chambers
- moved setup to a cabinet to have system in the "dark"
DH5α, DH5α/pUC19, and Shewie co-culture experiment setup
- Chamber 1: DH5α + wt Shewie + Lactose
- Chamber 2: DH5α + wt Shewie + Lactate (pos. control)
- Chamber 3: DH5α + wt Shewie (neg. control)
- Chamber 4: DH5α/pUC19 + wt Shewie + Lactose
- Chamber 5: DH5α/pUC19 + wt Shewie + Lactate (pos. control)
- Chamber 6: DH5α/pUC19 + wt Shewie (neg. control)
- Chamber 7: wt Shewie + Lactose (neg. control)
- Chamber 8: DH5α + Lactose (neg. control)
- Chamber 9: DH5α/pUC19 + Lactose (neg. control)
Tuesday: August 12, 2008
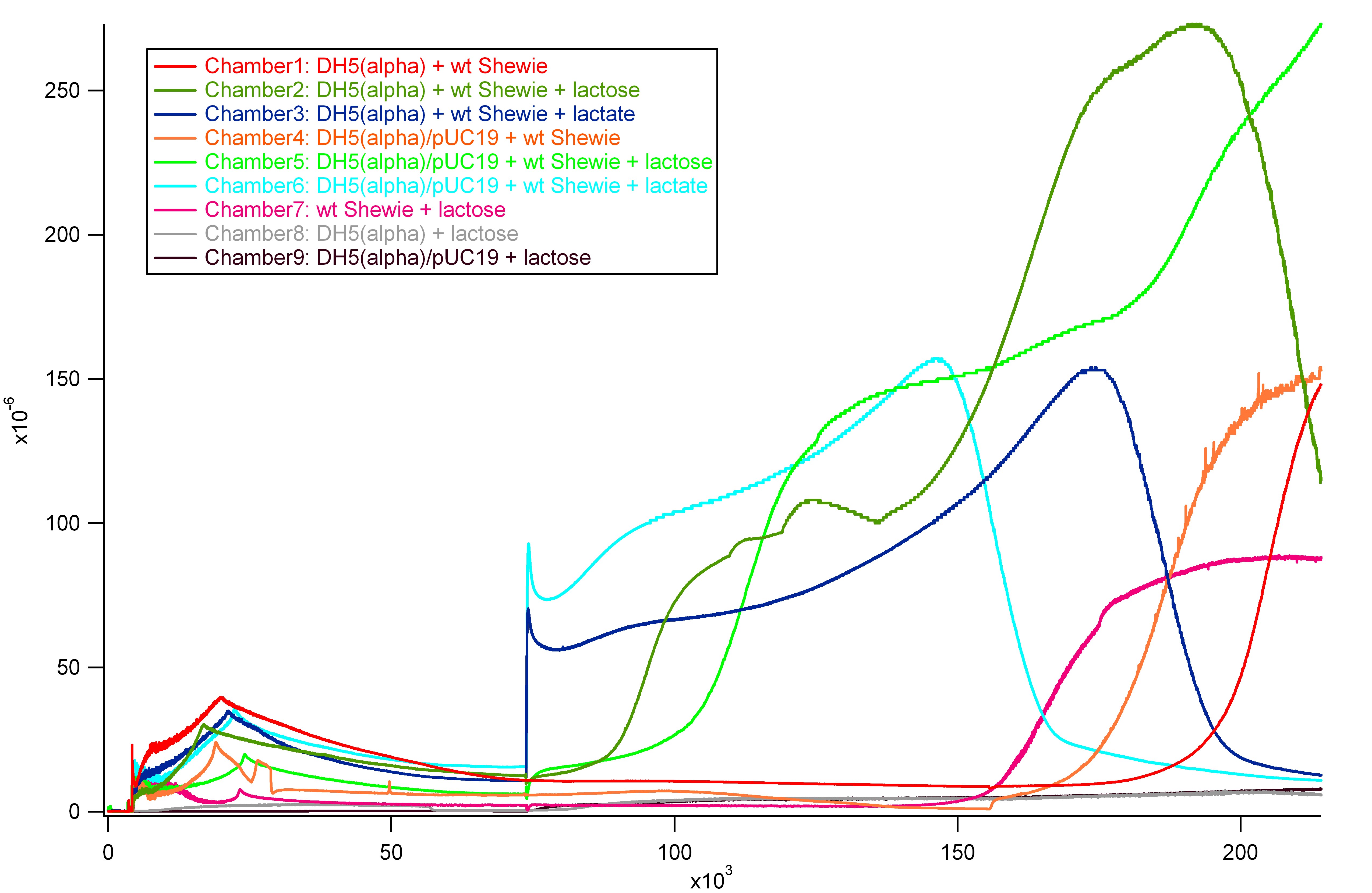
DH5(alpha) & DH5(alpha)/puC19 + wt Shewie Co-Culture Results
First Half
- Notes:
- At t = 5000 s, all cells added
- At t = 90000 s, all injections done
- At t = 155000 s, Lactose injections on Chambers 1 and 4
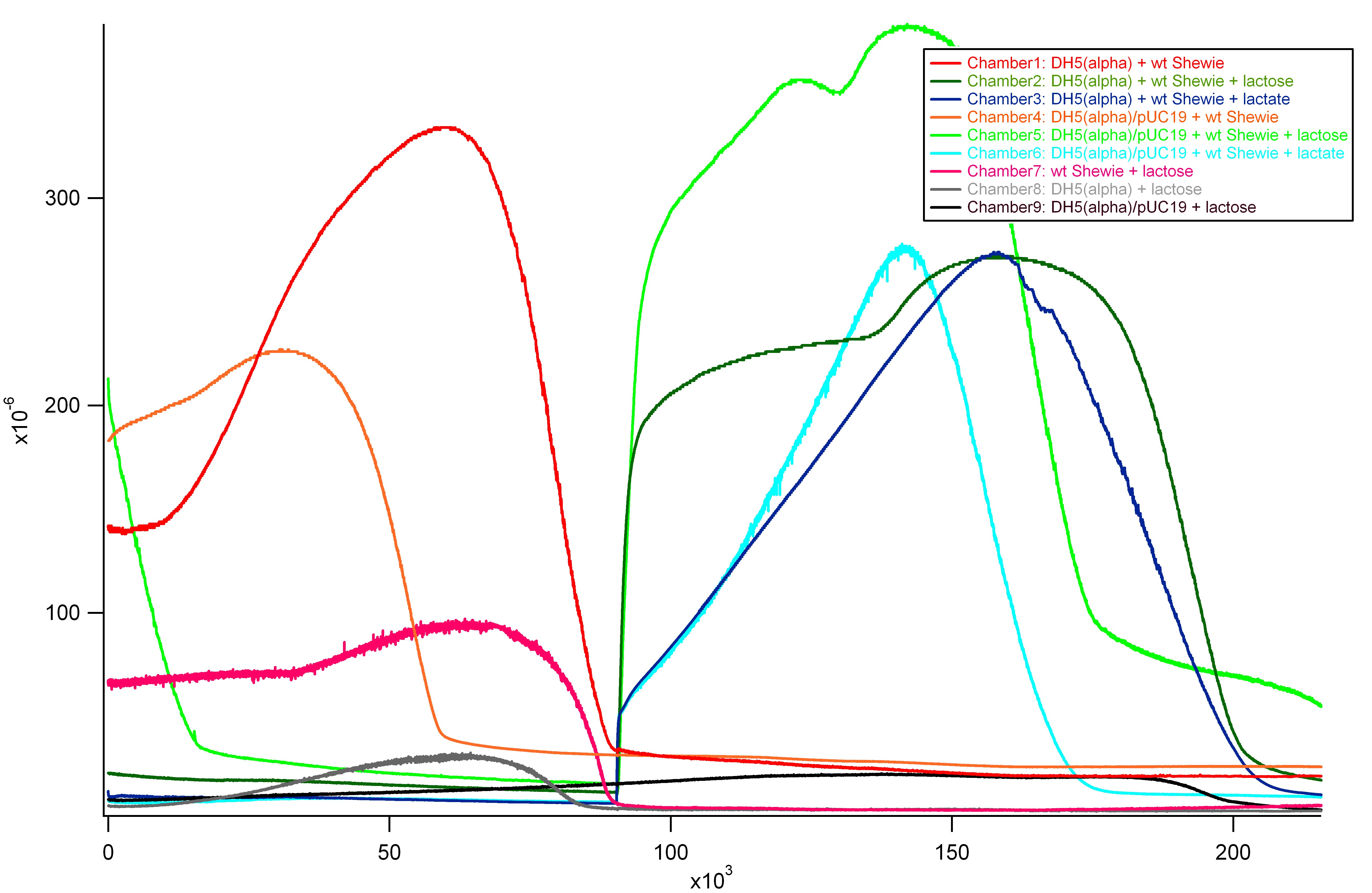
Second Half
- Notes:
- Computer restarted in middle of night, t=0 in second graph is 7 hours after end of first graph
- At t = 925000 s,
- Lactose injections on Chambers 2 and 5
- Lactate injections on Chambers 3 and 6
Concerns
- Repeatable current production in DH5(alpha) alone
- Possible contamination?
- Distinguishing features to tell current production of DH5(alpha) and DH5(alpha)/pUC19 apart
- After first lactose injection, the current production of the two have noticeable difference in slope
- However, initially after second lactose injection, the graphs overlap
Wednesday: August 13, 2008
RU1012 Light Repressible system experiment
- Streaked plates.
- All Cm/Amp plates with Xgal
- Dark
- Plate 1: RU1012 w/ plasmids
- Plate 2: RU1012
- Plate 3: Negative control (No bugs)
- Light
- Plate 1: RU1012 w/ plasmids
- Plate 2: RU1012
- Plate 3: Negative control (No bugs)
Results
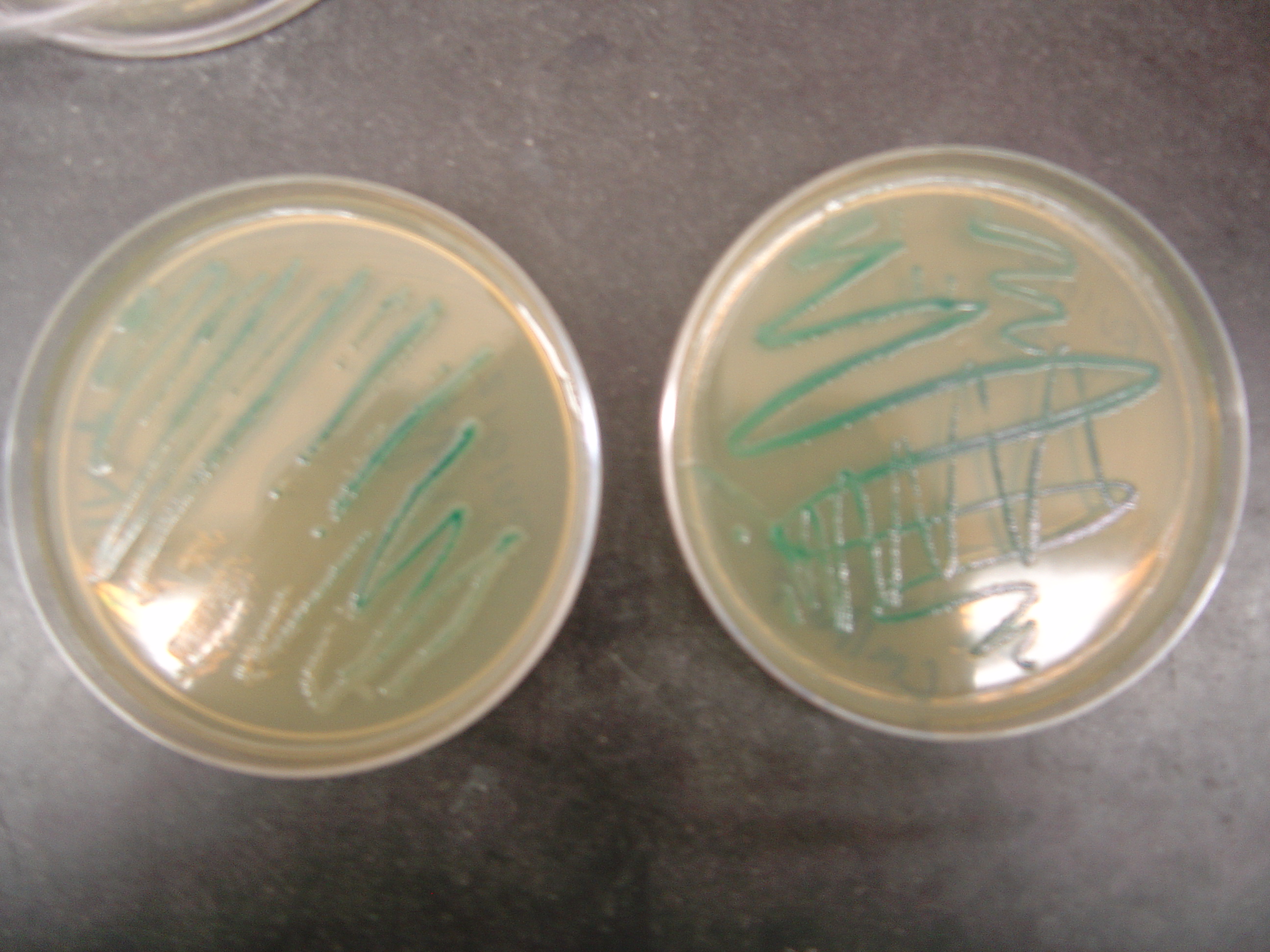
- RU1012 with Plasmid
- Notes:
- Plate on left was grown under light
- Plate on right was grown in the dark
- Possible repression seen in bottom left corner of the plate, but seems leaky
- Notes:
- RU1012
- Notes:
- Plate on left was grown under light
- Plate on right was grown in the dark
- Exhibited same behavior as RU1012 with plasmid
- Notes:
- Negative Control
- Nothing on plates for both light and dark
- Conclusions
- Hard to distinguish if it worked with streaked colonies
- May work better with lawns
Co-Culture Experiment Follow-up
- When disassembling the fuel cells, took the chamber media and spread them on Xgal plates
- Way of determining if contamination occured
- Controls are not expected to turn blue
Thursday: August 14, 2008
RU1012 Light Repressible system experiment
- Spread plates to try to create lawn.
- All Cm/Amp plates with Xgal
- Dark
- Plate 1: RU1012 w/ plasmids
- Plate 2: RU1012
- Plate 3: Negative control (No bugs)
- Light
- Plate 1: RU1012 w/ plasmids
- Plate 2: RU1012
- Plate 3: Negative control (No bugs)
Results
- Conclusion
- Possibly light is too strong? Explaining no growth in middle of plate grow under light
Friday: August 15, 2008
RU1012 Light Repressible system experiment
- Repeated previous day's experiment exactly
- Placed light further away
Co-culture Experiment Follow-up
- All plates turned blue
- Control chambers very likely were contaminated
- Will explore possibilities of autoclaving chambers
 "
"