Team:Tokyo Aplication project
From 2008.igem.org
| Line 6: | Line 6: | ||
<div> </div> | <div> </div> | ||
| - | {| style="color:#1b2c8a;background-color:#0c6;" cellpadding="3" cellspacing="1" border="1" bordercolor="#fff" width=" | + | {| style="color:#1b2c8a;background-color:#0c6;" cellpadding="3" cellspacing="1" border="1" bordercolor="#fff" width="60%" align="center" |
| - | !align="center" width= | + | !align="center" width=20% style="text-align:center"|[[Team:Tokyo_Tech/Parts|Parts Submitted to the Registry]] |
| - | !align="center" width= | + | !align="center" width=20% style="text-align:center"|[[Team:Tokyo_Tech/Team|Our Team]] |
| - | !align="center" width= | + | !align="center" width=20% style="text-align:center"|[[Team:Tokyo_Tech/Acknowledgements|Acknowledgements]] |
|} | |} | ||
Latest revision as of 16:19, 26 October 2008
| Basic project | Aplication project |
|---|
| Parts Submitted to the Registry | Our Team | Acknowledgements |
|---|
Contents |
Aplication project
|
About one paper, 94 times activity was observed at 30MPa for the lac
promoter compared with the level expressed at atmospheric pressure.
(cf.Takako Sato, Chiaki Kato,and Koki Horikoshi(1994) Effect of high
pressure on gene expression by lac and tac promoters in Escherichia
coli.Marine Biotechnology 3:89-92)
|
Development of low pressure inducible promoter
Method
Principle
LacI binds to lacI binding site and repress lac promoter. In addition, pressure of 30 MPa activated the lac promoter. We propose a hypothesis that 30 MPa induce a conformational change in lacI reprssure and change the affinity for lacI binding site weaker. Therefore, lacI can't repress lac promoter under 30 MPa.
Strategy
We are trying to develop low pressure inducible promoter by PCR random mutagenesis to lac promoter in order to reduce affitity to lacI. And we are screening an E. coli library for promoters that are induced under low pressure using a fluorescence activated cell sorter (FACS). This scheme is based on the ability to separate bacteria with a FACS in response to expression, or lack of expression, of a fluorescent marker.
- step 1 - Fluorescent bacteria without repressor protein were collected by FACS. This sorted pool contains bacteria bearing both constitutive and low pressure-inducible promoter.
- step 2 - Constitutive promoter are removed with repressor protein and sorting all non-fluorescent bacteria.
- step 3 - A final passage through under pressure with repressor protain and sorting for fluorescent bacteria removes false negatives and enriches for bacteria bearing promoter that are low pressure inducible.
Results
We finished step 1. Fluorescent and non-fluorescent bacteria were sorted and we characterized their promoter.
Sequence and Characterization
We sorted fluorescent (A) and non-fluorescent bacteria (B) with a FACS. Then, we analyze these base sequences.
- A have mutations in lacI binding site or non-functional DNA.
- B have mutations in CAP binding site, -35 or non-functional DNA.
Conclusion
We have successfully demonstrated that it is possible to collect objective promoter by PCR random mutagenesis and screening with a FACS. So we clearly believe that we can screen low pressure inducible lac promoter mutant with this strategy.
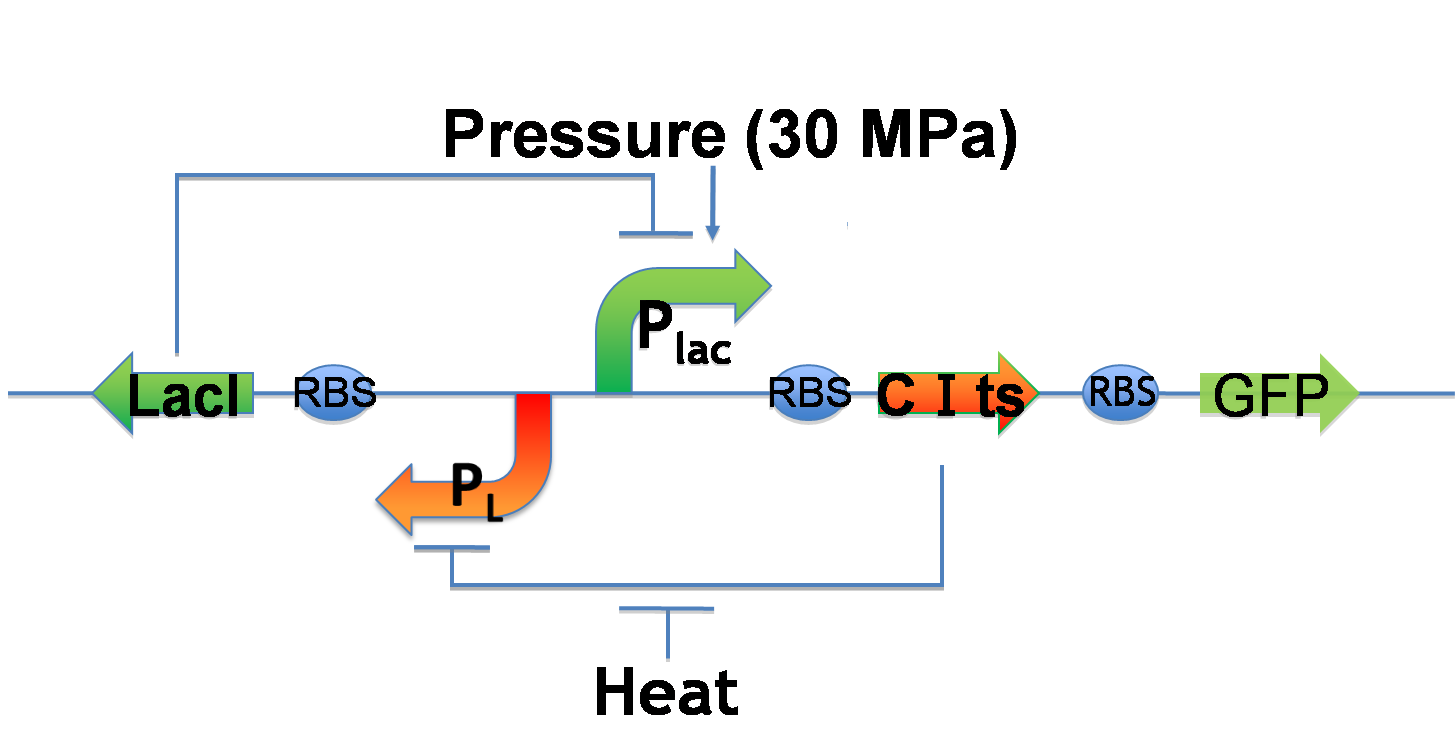
Genetic toggle switch to implement rewritable function
In order to implement rewritable function, we tried to construct Pressure Inducible Genetic Toggle Switch (in short PIGTS). We thought two types of model - genetic circuit model and mathematical model - so as to construct PIGTS.
Genetic circuit model
1. Writable function
- Plac is induced under 30MPa pressure
- Express CI and GFP
- CI represses PL and causes low LacI concentration
- Express increasingly CI and GFP(inserted destruction tags) ⇒ Bright!!
2. Erasable function
- PL is induced by heat
- Express LacI and LacI represses Plac
- Therefore, GFP expression decreases
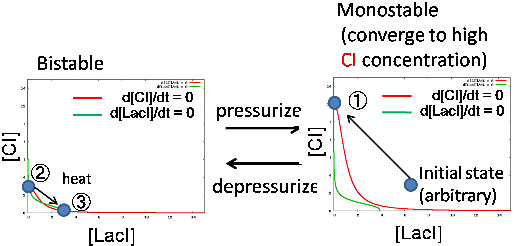
Mathematical model
We used two differential equation models and we call them 'atmospheric pressure model' and '30MPa pressure model'. In order to implement rewritable function, we decided to use the difference of nullclines of each system so as to satisfy the following condition
- Converge to high CI concentration state under 30MPa pressure (writable)
- Bistable under atmospheric pressure (erasable)
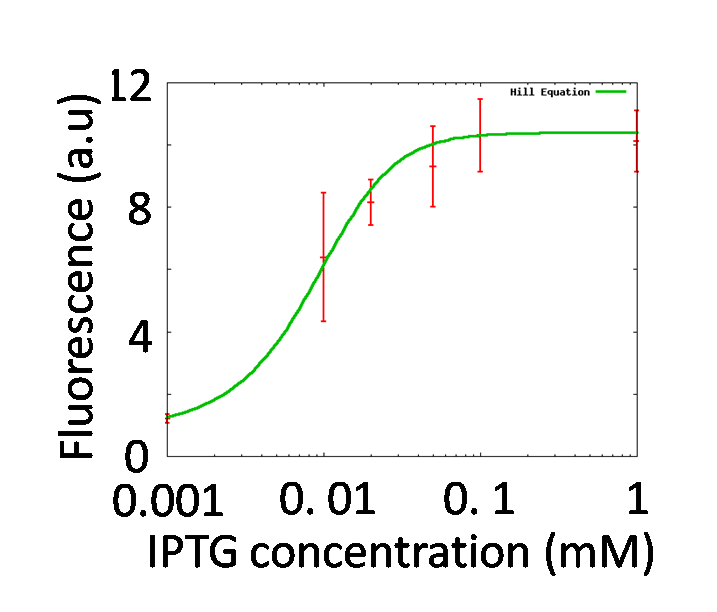
IPTG assay
Objective
By testing how LacI represses the lac promoter, Hill coefficient of lac promoter should be decided. In order to adjust effective concentration of LacI, IPTG was added.
Result & Conclusion
GFP fluorescence intensity was enhanced in an IPTG-dose dependent manner. It indicates that the LacI repression was getting weak by adding IPTG. Taken together with the graph shown in figure 1 and the formulation of Hill function fitting described above, the characteristics of the lac promoter expressed in Hill function was determined. Finally, we obtained nPlac = 2.15.
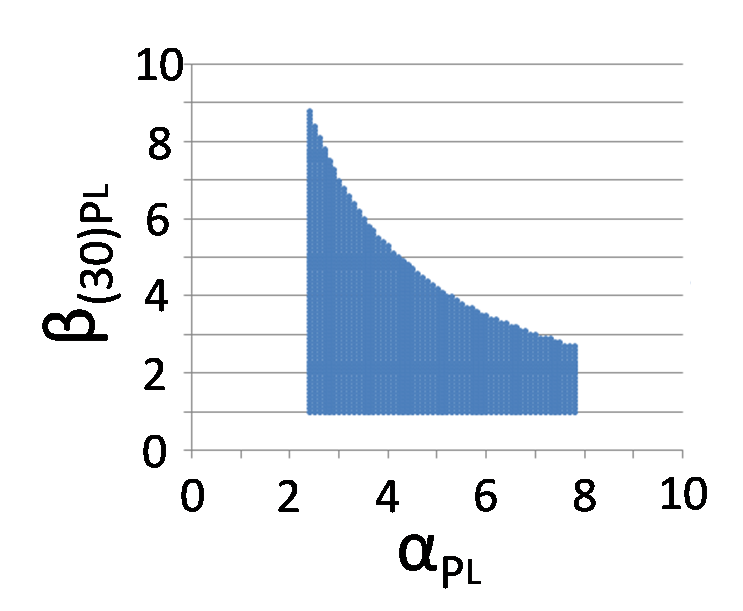
Calculate the domain of appropriate parameters
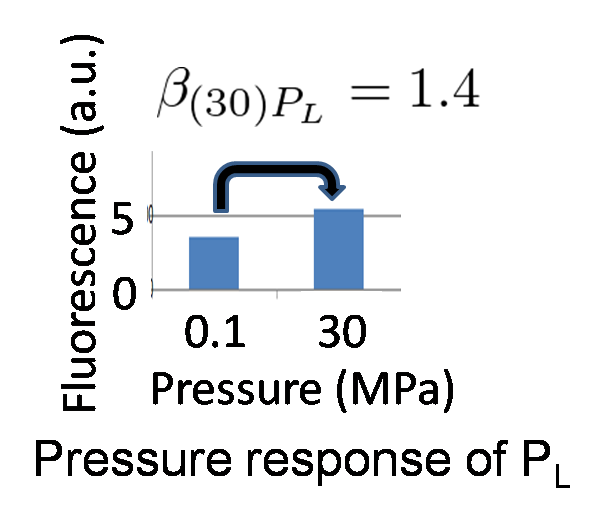
It is known that Plac activity under 30MPa pressure is 94.0 times stronger than Plac activity under atmospheric pressure, so, β30Plac = 94.0. If 30MPa additional activity rate of PL (β30PL) or the ratio of Plac - RBS strength (αPlac) and PL-RBS strength(αPL) are not appropriate value, we can't construct PIGTS. So we calculated the domain of appropriate β30PL and the ratio of αPlac and αPL.
We identified β30PL = 1.42 by our experiment under 30MPa pressure(figure 5-6). Therefore, we can implement rewritable function if we choose PL - RBS strength range from 2.4 to 7.4.
 "
"