Team:Freiburg Transfection
From 2008.igem.org
(Difference between revisions)
| Line 5: | Line 5: | ||
<br> | <br> | ||
<br> | <br> | ||
| + | <h3>'''1) Testing the transfection protocol'''</h3> | ||
| + | In order to check if the transfection protocol is suitable to transfect 293T cells a 'test'transfection with a lac z gene was done and the β-galactosidase was detected with an ONPG test.<br> | ||
| + | ONPG-assay: <br> | ||
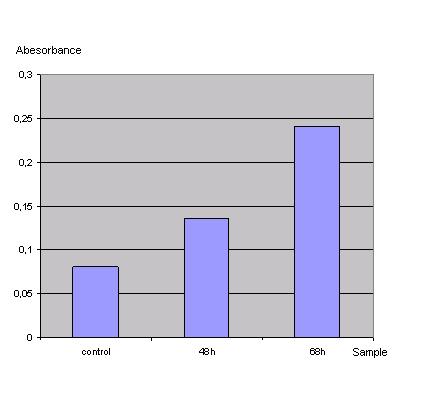
| + | <br>[[Image:FreiGEMTable3onpg.JPG]]<br> | ||
| + | ''Table3: Absorbance of o-Nitrophenol produced by the β-galactosidase'' | ||
| + | <br><br> | ||
| + | [[Image:Graph2onpg.JPG]]<br> | ||
| + | ''Graph2: Absorbance of o-Nitrophenol produced by the β-galactosidase'' | ||
| + | Control was done with untransfected cells using the same procedure.<br> | ||
| + | <br> | ||
| + | <h3>'''2) Testing the transfectionvector-CMV promoter construct'''</h3> | ||
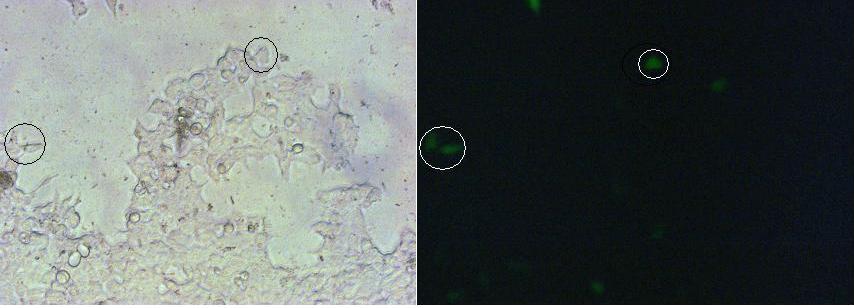
| + | To test the functionality of the transfectionvector and the CMV-promoter, CFP and YFP was cloned behind the promoter and the plasmid was brought into 293T cells.<br> | ||
| + | The transfected cells show fluorescence by excitation of 510-520nm while the untransfected remain dark at this wavelength <br> | ||
| + | [[Image:FreigemCostructtv.JPG|800px]]<br> | ||
| + | ''Picture1:Transfected cells without (left) and with YFP-filter (right)'' | ||
| + | <br> | ||
| + | |||
<h3>'''1) Localization at the cell membrane'''</h3> | <h3>'''1) Localization at the cell membrane'''</h3> | ||
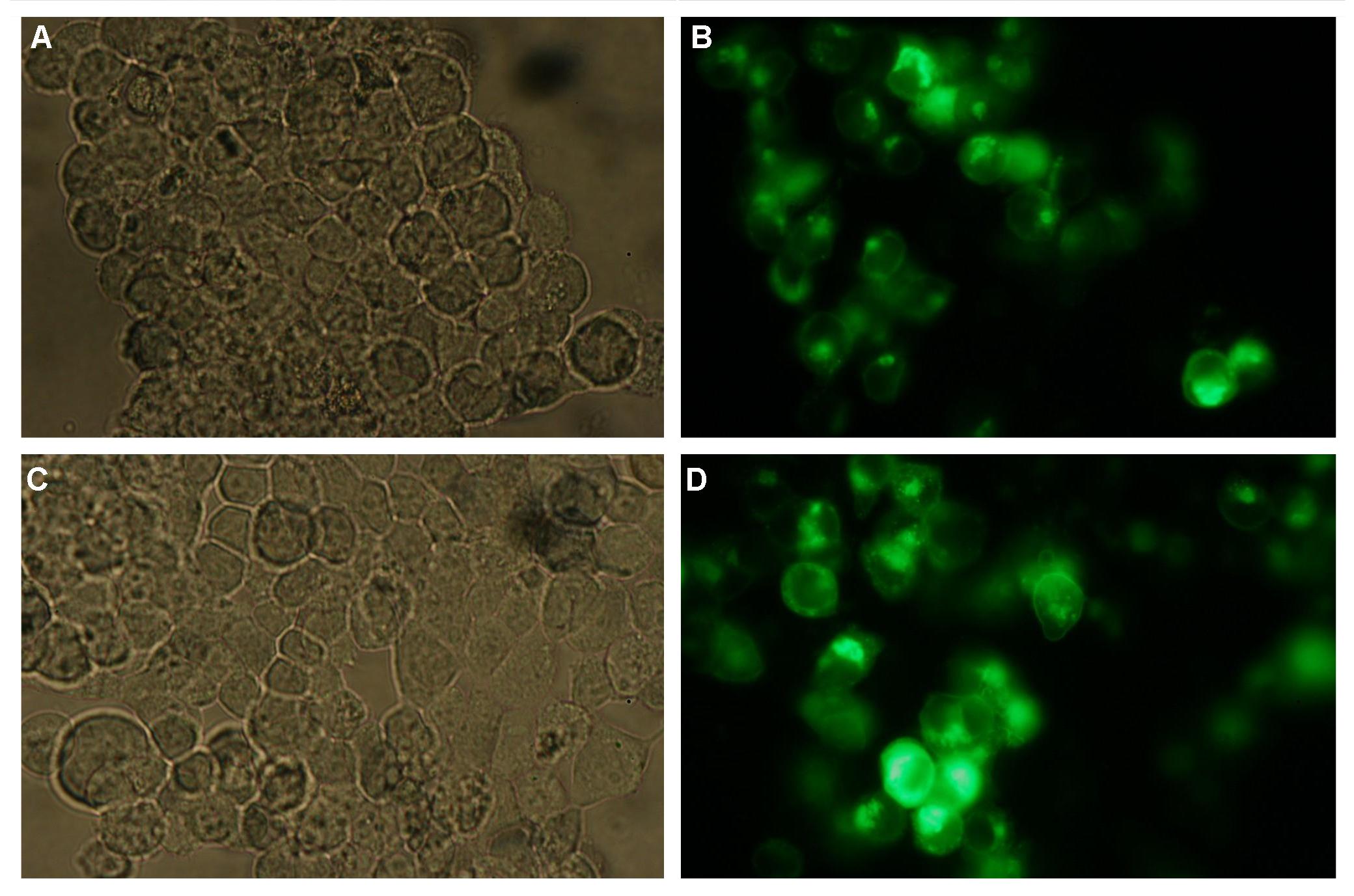
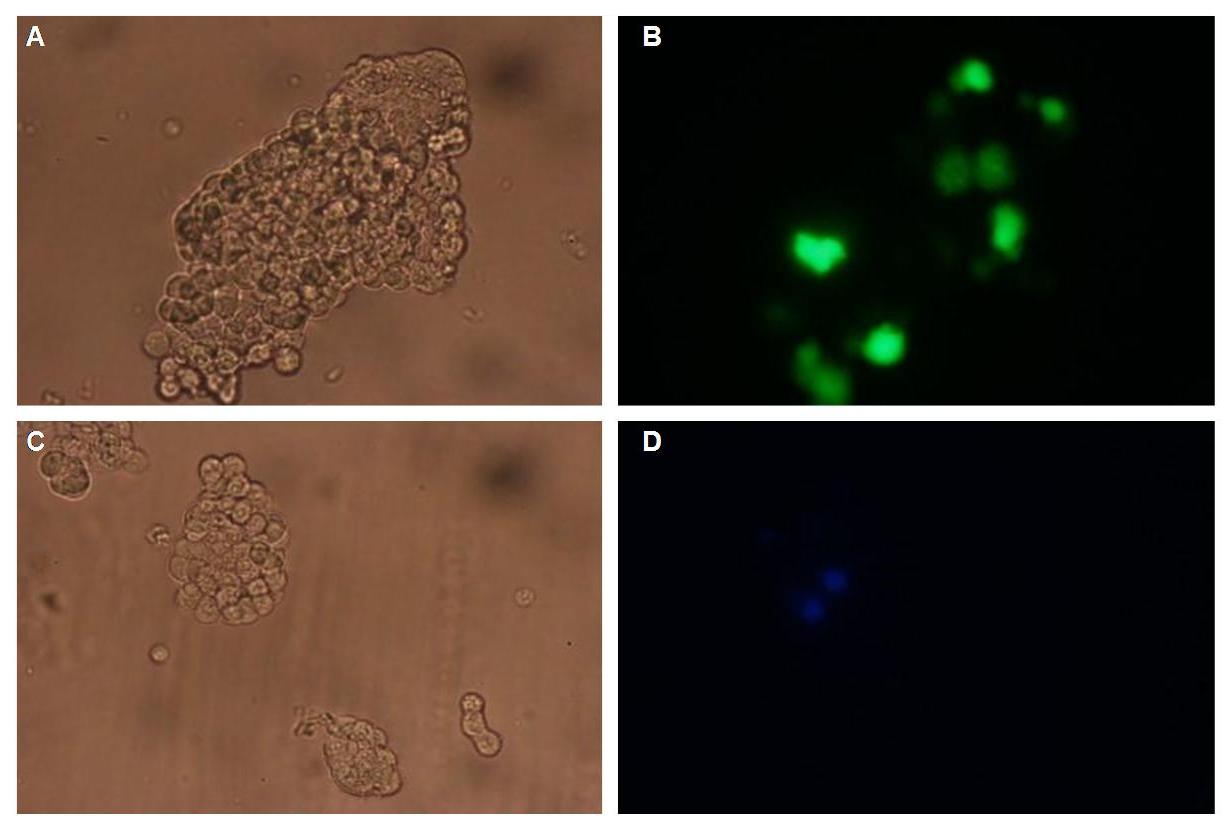
To show the localization of the constructs at the cell membrane transfection of the construct signalpeptide-Lipocalin-transmembraneregion-betaLactamase1-YFP was performed.<br> | To show the localization of the constructs at the cell membrane transfection of the construct signalpeptide-Lipocalin-transmembraneregion-betaLactamase1-YFP was performed.<br> | ||
| Line 36: | Line 53: | ||
'''Figure 5_Transfection'''<br> | '''Figure 5_Transfection'''<br> | ||
<br> | <br> | ||
| - | |||
| - | + | ||
| - | + | ||
| + | |||
<br>The detection of YFP took place 1 day later under a microscope with YFP filter.<br> | <br>The detection of YFP took place 1 day later under a microscope with YFP filter.<br> | ||
<br> | <br> | ||
'''Ca2+ precipitation'''<br><br> | '''Ca2+ precipitation'''<br><br> | ||
Transfection of the trasfectionvector+cmv+YFP: <br> | Transfection of the trasfectionvector+cmv+YFP: <br> | ||
| - | The transfected cells | + | The transfected cells show fluorescence by excitation of 510-520nm while the untransfected remain dark at this wavelength <br> |
[[Image:FreigemCostructtv.JPG|800px]]<br> | [[Image:FreigemCostructtv.JPG|800px]]<br> | ||
''Picture1:Transfected cells without (left) and with YFP-filter (right)'' | ''Picture1:Transfected cells without (left) and with YFP-filter (right)'' | ||
<br> | <br> | ||
<br><br> | <br><br> | ||
| - | + | ||
| - | + | ||
| - | '' | + | |
| - | <br | + | '''Ca2+ precipitation:'''<br<<br> |
| - | + | Due to the results of the ONPG assay the Ca2+ precipitation proofed to be a very simple and effective method to transfect the 293t cells.<br> | |
| - | + | The transfection with our transfectionvector + CMV + YFP showed that this construct works well enough to be cloned together with our artificial receptors.<br> | |
| - | + | ||
'''METHODS''' | '''METHODS''' | ||
_________________________________________________________________________________________________________________<br><br> | _________________________________________________________________________________________________________________<br><br> | ||
'''Transfection of 293T cells'''<br> | '''Transfection of 293T cells'''<br> | ||
| - | One day before transfection cells were counted in the Neubauer chamber and 6*10^4 cells/cm² were seeded in 6 well plates. Approximately 1 hour before transfection cells were washed with 1xPBS and fresh DMEM medium was added. For transfection 2µg of DNA were mixed with 25µl CaCl2 and ddH2O was filled up to 250µl. After an incubation on ice for 20 min 250µl BBS (2x) were added. This mixture was given to the cells and after 4-12 hours cells were washed and fresh medium was added. | + | One day before transfection cells were counted in the Neubauer chamber and 6*10^4 cells/cm² were seeded in 6 well plates. Approximately 1 hour before transfection cells were washed with 1xPBS and fresh DMEM medium was added. For transfection 2µg of DNA were mixed with 25µl CaCl2 and ddH2O was filled up to 250µl. After an incubation on ice for 20 min 250µl BBS (2x) were added. This mixture was given to the cells and after 4-12 hours cells were washed and fresh medium was added.<br> |
| - | + | '''ONPG Test'''<br> | |
| + | Transfection was performed with a lac z gene using the transfection protocol described above. After 48h one part of the cells was harvested by washing them in PBS and scraping them off. Then the cells were centrifuged at 13000rpm for 2 min and the PBS was replaced by 500µl lysisbuffer (1x). Incubation took place at -80°C for 20min. After thawing the solution was vortexed, spun down and the supernatant was frozen at -20°C. The same procedure was done with the rest of the cells one day later (68h). Then 20µl of each lysate was given to 130µl reactionbuffer (incl. ONPG) letting the mixture incubate for 1h at 37°C. Measurement was done using the ELISA-reader at 405nm. | ||
Revision as of 17:10, 28 October 2008
 "
"