Team:Chiba/Calendar-Home/21 August 2008
From 2008.igem.org
(Difference between revisions)
(New page: '''OUTPUT TEAM''' *transformation Lux CDABE(J32007:2007),RBS(B0034:2007) :結果:成功→RBS :失敗→Lux CDABE(二回行ったがどちらもコロニーができなかった)→三...) |
(→Team:Communication) |
||
| (23 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
| - | + | >[[Team:Chiba|Home]] | [[Team:Chiba/Notebook|Notebook]] | |
| - | + | ||
| - | + | ||
| - | : | + | [[Team:Chiba/Calendar-Home/20 August 2008|20 August 2008 <]]|[[Team:Chiba/Calendar-Home/22 August 2008|> 22 August 2008]] |
| - | : | + | |
| + | ==Laboratory work== | ||
| + | ===Team:Input=== | ||
| + | (-->20/8) | ||
| + | |||
| + | Inoculated transformants for 12 hours in LB 2mL containing Ampicillin. | ||
| + | |||
| + | *[http://partsregistry.org/Part:BBa_R0051 BBa_R0051](2007) | ||
| + | *[http://partsregistry.org/Part:BBa_R0051 BBa_R0051](2006) | ||
| + | *[http://partsregistry.org/Part:BBa_J06650 BBa_J06650](2007) | ||
| + | *[http://partsregistry.org/Part:BBa_J06650 BBa_J06650](2006) | ||
| + | *[http://partsregistry.org/Part:BBa_J22136 BBa_J22136](2007) | ||
| + | *[http://partsregistry.org/Part:BBa_J22141 BBa_J22141](2007) | ||
| + | |||
| + | BBa_J22141:After inoculated for 11 hours, added 20μl IPTG and cultured one hour. | ||
| + | |||
| + | |||
| + | *UV irradiation test (plate phase) | ||
| + | *two plasmids from(JW1908) | ||
| + | **(Repressor)-Ptrc-LuxR-Plux-cI-colE1-Amp- | ||
| + | **(Reporter)-PcI-GFP-p15a-Cm- | ||
| + | |||
| + | #Inoculated (Reporter) culture from glycerol stock(-80°C freezer) in 2mL LB-Ampicillin, Chloramphenicol medium and LB-Ampicillin, Chloramphenicol, 100nmAHL mediumfor 12h, at 37°C. | ||
| + | #We diluted 10<sup>5</sup>-fold, and plated 20ul of the resulting solution to (LB-Amp,Cm)(LB-Amp,cm,AHL100nM) plates each other. | ||
| + | #We cultured for 12h at 37°C. | ||
| + | #We iraddiated UV each plate. Wavelength:254nm,distance from the UV lamp to the cultures were 14cm. | ||
| + | #We performed the same operations on plates following 9 or 21h of UV | ||
| + | exposure. | ||
| + | #Colonies from both plates were picked and cultured in 2mL of | ||
| + | LB-Amp,Cm or LB-Amp, Cm, 100nMAHL for 12h at 37°C. | ||
| + | |||
| + | #We diluted 10<sup>5</sup>-fold, and plated 20ul of the | ||
| + | resulting solution to (LB-Amp,Cm)(LB-Amp,cm,AHL100nM) plates each other. | ||
| + | |||
| + | |||
| + | *Colony count | ||
| + | <table width="200" border="4" cellpadding="0" cellspacing="0" bordercolor="#000000"> | ||
| + | <td width="257"></td> | ||
| + | <td>no AHL</td><td>AHL100nM</td> | ||
| + | <tr> | ||
| + | <td>9h</td> | ||
| + | <td>606</td><td>453</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>12h</td> | ||
| + | <td>146</td><td>151</td> | ||
| + | </tr> | ||
| + | </table> | ||
| + | result | ||
| + | <BR> | ||
| + | GFP fluorescence was observed from plates without AHL. | ||
| + | |||
| + | ===Team:Communication=== | ||
| + | :(20/8)--->'''[[Team:Chiba/protocol/PCR|PCR]]''' | ||
| + | ::*[http://partsregistry.org/Part:BBa_C0070 BBa_C0070](2007) | ||
| + | ::*[http://partsregistry.org/Part:BBa_C0070 BBa_C0070](2006) | ||
| + | ::*[http://partsregistry.org/Part:BBa_C0076 BBa_C0076](2007) | ||
| + | ::*[http://partsregistry.org/Part:BBa_C0078 BBa_C0078](2007) | ||
| + | ::*[http://partsregistry.org/Part:BBa_C0078 BBa_C0078](2006) | ||
| + | |||
| + | |||
| + | <table width="315" border="2" cellpadding="0" cellspacing="0" bordercolor="#000000"> | ||
| + | <tr> | ||
| + | <td width="257">DNA Template</td> | ||
| + | <td>1</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>dNTP mix(μL)</td> | ||
| + | <td>10</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Foward Primer(μL)</td> | ||
| + | <td>5</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Reverse Primer(μL)</td> | ||
| + | <td>5</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>DNA polymerase TAQ(μL)</td> | ||
| + | <td>1</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Thermopol Buffer(μL)</td> | ||
| + | <td>5</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>dH<sub>2</sub>O(μL)</td> | ||
| + | <td>28</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>TOTAL(μL)</td> | ||
| + | <td>50</td> | ||
| + | </tr> | ||
| + | </table> | ||
| + | |||
| + | |||
| + | :95°C,5min -> ( 95°C,1min -> 52°C,1min -> 72°C,1min )・・・25cycles -> 72°C,10min -> 6°C | ||
| + | |||
| + | |||
| + | |||
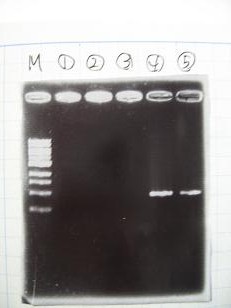
| + | :--->'''[[Team:Chiba/protocol/gelcheck|Gel Check]]''' | ||
| + | {|align="justify" | ||
| + | |[[Image:Chiba-0821.JPG]] | ||
| + | | | ||
| + | :<table width="315" border="2" cellpadding="0" cellspacing="0" bordercolor="#000000"> | ||
| + | <tr> | ||
| + | <td width="257">Sample DNA</td> | ||
| + | <td>3</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>Loading Dye(μL)</td> | ||
| + | <td>2</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>dH<sub>2</sub>O(μL)</td> | ||
| + | <td>7</td> | ||
| + | </tr> | ||
| + | <tr> | ||
| + | <td>TOTAL(μL)</td> | ||
| + | <td>12</td> | ||
| + | </tr> | ||
| + | </table> | ||
| + | |||
| + | :From left; | ||
| + | ::*[http://partsregistry.org/Part:BBa_C0078 BBa_C0078](2007) -> None | ||
| + | ::*[http://partsregistry.org/Part:BBa_C0078 BBa_C0078](2006) -> None | ||
| + | ::*[http://partsregistry.org/Part:BBa_C0076 BBa_C0076](2007) -> None | ||
| + | ::*[http://partsregistry.org/Part:BBa_C0070 BBa_C0070](2007) -> OK | ||
| + | ::*[http://partsregistry.org/Part:BBa_C0070 BBa_C0070](2006) -> OK | ||
| + | |} | ||
| + | |||
| + | |||
| + | |||
| + | :'''[[Team:Chiba/protocol/transformation|Transformation]]''' | ||
| + | :competent cells : XL10G | ||
| + | ::*[http://partsregistry.org/Part:BBa_S03154 BBa_S03154](2007) | ||
| + | ::*[http://partsregistry.org/Part:BBa_S03154 BBa_S03154](2006) | ||
| + | ::*[http://partsregistry.org/Part:BBa_I9026 BBa_I9026](2007) | ||
| + | ::*[http://partsregistry.org/Part:BBa_I9026 BBa_I9026](2006) | ||
| + | ::*[http://partsregistry.org/Part:BBa_I9030 BBa_I9030](2007) | ||
| + | ::*[http://partsregistry.org/Part:BBa_I9030 BBa_I9030](2006) | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | --->(23/8)'''[[Team:Chiba/protocol/digestion|Digestion]]''' | ||
| + | |||
| + | ===Team:Output=== | ||
| + | [[Team:Chiba/protocol/transformation|Transformation]] | ||
| + | *[http://partsregistry.org/Part:BBa_J32007 BBa_J32007](2007)-->no colony-->no colony(2 times)-->colony(3times) | ||
| + | *[http://partsregistry.org/Part:BBa_B0034 BBa_B0034](2007)-->colony | ||
Latest revision as of 05:58, 30 October 2008
20 August 2008 <|> 22 August 2008
Contents |
Laboratory work
Team:Input
(-->20/8)
Inoculated transformants for 12 hours in LB 2mL containing Ampicillin.
- [http://partsregistry.org/Part:BBa_R0051 BBa_R0051](2007)
- [http://partsregistry.org/Part:BBa_R0051 BBa_R0051](2006)
- [http://partsregistry.org/Part:BBa_J06650 BBa_J06650](2007)
- [http://partsregistry.org/Part:BBa_J06650 BBa_J06650](2006)
- [http://partsregistry.org/Part:BBa_J22136 BBa_J22136](2007)
- [http://partsregistry.org/Part:BBa_J22141 BBa_J22141](2007)
BBa_J22141:After inoculated for 11 hours, added 20μl IPTG and cultured one hour.
- UV irradiation test (plate phase)
- two plasmids from(JW1908)
- (Repressor)-Ptrc-LuxR-Plux-cI-colE1-Amp-
- (Reporter)-PcI-GFP-p15a-Cm-
- Inoculated (Reporter) culture from glycerol stock(-80°C freezer) in 2mL LB-Ampicillin, Chloramphenicol medium and LB-Ampicillin, Chloramphenicol, 100nmAHL mediumfor 12h, at 37°C.
- We diluted 105-fold, and plated 20ul of the resulting solution to (LB-Amp,Cm)(LB-Amp,cm,AHL100nM) plates each other.
- We cultured for 12h at 37°C.
- We iraddiated UV each plate. Wavelength:254nm,distance from the UV lamp to the cultures were 14cm.
- We performed the same operations on plates following 9 or 21h of UV
exposure.
- Colonies from both plates were picked and cultured in 2mL of
LB-Amp,Cm or LB-Amp, Cm, 100nMAHL for 12h at 37°C.
- We diluted 105-fold, and plated 20ul of the
resulting solution to (LB-Amp,Cm)(LB-Amp,cm,AHL100nM) plates each other.
- Colony count
| no AHL | AHL100nM | |
| 9h | 606 | 453 |
| 12h | 146 | 151 |
result
GFP fluorescence was observed from plates without AHL.
Team:Communication
- (20/8)--->PCR
- [http://partsregistry.org/Part:BBa_C0070 BBa_C0070](2007)
- [http://partsregistry.org/Part:BBa_C0070 BBa_C0070](2006)
- [http://partsregistry.org/Part:BBa_C0076 BBa_C0076](2007)
- [http://partsregistry.org/Part:BBa_C0078 BBa_C0078](2007)
- [http://partsregistry.org/Part:BBa_C0078 BBa_C0078](2006)
| DNA Template | 1 |
| dNTP mix(μL) | 10 |
| Foward Primer(μL) | 5 |
| Reverse Primer(μL) | 5 |
| DNA polymerase TAQ(μL) | 1 |
| Thermopol Buffer(μL) | 5 |
| dH2O(μL) | 28 |
| TOTAL(μL) | 50 |
- 95°C,5min -> ( 95°C,1min -> 52°C,1min -> 72°C,1min )・・・25cycles -> 72°C,10min -> 6°C
- --->Gel Check
- Transformation
- competent cells : XL10G
- [http://partsregistry.org/Part:BBa_S03154 BBa_S03154](2007)
- [http://partsregistry.org/Part:BBa_S03154 BBa_S03154](2006)
- [http://partsregistry.org/Part:BBa_I9026 BBa_I9026](2007)
- [http://partsregistry.org/Part:BBa_I9026 BBa_I9026](2006)
- [http://partsregistry.org/Part:BBa_I9030 BBa_I9030](2007)
- [http://partsregistry.org/Part:BBa_I9030 BBa_I9030](2006)
--->(23/8)Digestion
Team:Output
- [http://partsregistry.org/Part:BBa_J32007 BBa_J32007](2007)-->no colony-->no colony(2 times)-->colony(3times)
- [http://partsregistry.org/Part:BBa_B0034 BBa_B0034](2007)-->colony
 "
"