Team:Paris/Notebook/Protocols
From 2008.igem.org
(→Concentration of the Miniprep) |
(→PCR Screening) |
||
| (16 intermediate revisions not shown) | |||
| Line 61: | Line 61: | ||
* 30 µL of DNA + 6 µL of Loading Dye | * 30 µL of DNA + 6 µL of Loading Dye | ||
| - | '''Separate each | + | '''Separate each sample by an empty well !''' |
= Extraction = | = Extraction = | ||
| Line 68: | Line 68: | ||
* Use a new blade for each extraction | * Use a new blade for each extraction | ||
* The band weight must be less than 400 mg | * The band weight must be less than 400 mg | ||
| + | |||
| + | |||
| + | = Antarctic phosphatase = | ||
| + | To avoid autoligation, the vector in a construction has to be treated with phosphatase to dephosphorylate the cut end of the plasmid. The Antarctic phosphatase has the advantage to be easily and totally inactivated by heating. | ||
| + | * After the digestion reaction add Antarctic phosphatase buffer to a final concentration of 1x | ||
| + | * Add 1µL of Antarctic phosphatase for 1 to 5 µg of DNA | ||
| + | * Incubate at 37°C for 15 minutes and then inactivate the phosphatase at 65°C for 5 min | ||
| + | |||
| + | |||
| + | '''Do not use this reaction for the insert or the ligation will not work''' | ||
= Amplification of promoters = | = Amplification of promoters = | ||
| Line 100: | Line 110: | ||
== Gel Slice and PCR Product Preparation == | == Gel Slice and PCR Product Preparation == | ||
=== Dissolving the Gel Slice === | === Dissolving the Gel Slice === | ||
| - | * Following electrophoresis, excise DNA band from gel slice in a pre-weighed | + | * Following electrophoresis, excise DNA band from gel slice in a pre-weighed 2 mL microcentrifuge tube. |
* Add 10µL membrane Binding Solution per 10 mg of gel slice. We prefer not to vortex and we incubate at 50-65°C until gel slice is completely dissolved (∼10 min). Quick centrifuge. | * Add 10µL membrane Binding Solution per 10 mg of gel slice. We prefer not to vortex and we incubate at 50-65°C until gel slice is completely dissolved (∼10 min). Quick centrifuge. | ||
| Line 124: | Line 134: | ||
= Quantification by Electrophoresis = | = Quantification by Electrophoresis = | ||
* Gel : 1.5-2% agarose with BET added (5 µL BET for 100 mL TBE) | * Gel : 1.5-2% agarose with BET added (5 µL BET for 100 mL TBE) | ||
| - | * 10 µL Quick-Load 1 kb DNA Ladder | + | * 10 µL Quick-Load 100pb or 1 kb DNA Ladder |
* 2 µL Loadind Dye + 3 µL DNA | * 2 µL Loadind Dye + 3 µL DNA | ||
| Line 159: | Line 169: | ||
* LB medium: Dissolve 25mg/L of Luria Broth in purified water. Autoclave the solution. Store at room temperature and take care of any contamination | * LB medium: Dissolve 25mg/L of Luria Broth in purified water. Autoclave the solution. Store at room temperature and take care of any contamination | ||
| - | * LBA medium: Dissolve and gently mix with a magnetic barrel 40mg/L of Luria Broth Agar in purified water. Autoclave the solution. Store at room temperature and take care of any contamination. To make some plates | + | * LBA medium: Dissolve and gently mix with a magnetic barrel 40mg/L of Luria Broth Agar in purified water. Autoclave the solution. Store at room temperature and take care of any contamination. To make some plates heat the solidified medium with microwave. Wait untill you can hold the bottle in your hands and then add the appropriate antibiotic to a final concentration of 1x. Provide approximatively 20mL per plate in a sterile hood. Returned the plates and store them at +4°C |
= Minimum Medium = | = Minimum Medium = | ||
| Line 173: | Line 183: | ||
<br>- H<sub>2</sub>O QSP 200 mL | <br>- H<sub>2</sub>O QSP 200 mL | ||
| - | Filter the medium with a cell-culture unit of filtration. If needed add the appropriate antibiotic to a final concentration of | + | Filter the medium with a cell-culture unit of filtration. If needed add the appropriate antibiotic to a final concentration of 1x |
| + | |||
| + | = Soft Agar Medium = | ||
| + | |||
| + | * Add 3.5 to 4g of agar per liter of LB or Minimum Medium. Autoclave the solution. Store at room temperature and take care of any contamination. To make some plates heat the solidified medium with microwave. Wait untill you can hold the bottle in your hands and then add the appropriate antibiotic to a final concentration of 1x. Provide approximatively 20mL per plate in a sterile hood. Returned the plates and store them at +4°C | ||
| - | = | + | = 1000x stock antibiotic = |
| - | * Chloramphenicol: dissolve 30mg/mL of chloramphenicol in | + | * Chloramphenicol: dissolve 30mg/mL of chloramphenicol in ethanol 100%. Store at room temperature |
| - | * Tetracyclin: dissolve 12,5mg/mL of tetracyclin in a 50/50 | + | * Tetracyclin: dissolve 12,5mg/mL of tetracyclin in a 50/50 ethanol/water mix. Store at -20°C |
* Ampicillin: dissolve 100mg/mL of ampicillin in water. Filter the solution. Store at +4°C | * Ampicillin: dissolve 100mg/mL of ampicillin in water. Filter the solution. Store at +4°C | ||
* Kanamycin: dissolve 100mg/mL of kanamycin in water. Filter the solution. Store at room temperature | * Kanamycin: dissolve 100mg/mL of kanamycin in water. Filter the solution. Store at room temperature | ||
| Line 190: | Line 204: | ||
'''After''', add | '''After''', add | ||
* 12.5 µL Mix | * 12.5 µL Mix | ||
| - | * 0.5 µL Oligo | + | * 0.5 µL Oligo VF (10µM) |
| - | * 0.5 µL Oligo | + | * 0.5 µL Oligo VR (10µM) |
* 11,5 µL pure water | * 11,5 µL pure water | ||
*negative control : without clone's colony | *negative control : without clone's colony | ||
| Line 425: | Line 439: | ||
| | | | ||
|- style="text-align: center;" | |- style="text-align: center;" | ||
| - | | | + | |pflgB |
|yes | |yes | ||
|FlhDC | |FlhDC | ||
| Line 486: | Line 500: | ||
* Unfreeze strains from the glycerol stock containing or not the plasmid with a contitutive promoter and a fluorescent reporter gene | * Unfreeze strains from the glycerol stock containing or not the plasmid with a contitutive promoter and a fluorescent reporter gene | ||
| - | * Put a sterile tip in the 1.5 mL stock tube and then place it in a 50 mL Falcon with 5 ml of minimum medium. Over Night (16h) culture at 37°C / 300 rpm | + | * Put a sterile tip in the 1.5 mL stock tube and then place it in a 50 mL Falcon with 5 ml of [https://2008.igem.org/Team:Paris/Notebook/Protocols#Minimum_Medium minimum medium]. Over Night (16h) culture at 37°C / 300 rpm |
* Measure the OD<sub>600</sub> (Linear Zone Measurement <1.5) | * Measure the OD<sub>600</sub> (Linear Zone Measurement <1.5) | ||
* Dilute the culture medium in the same new medium to obtain an approximative OD<sub>600</sub> of 0.01 (V<sub>final</sub>= 50 mL in a 250 ml erlenmeyer) | * Dilute the culture medium in the same new medium to obtain an approximative OD<sub>600</sub> of 0.01 (V<sub>final</sub>= 50 mL in a 250 ml erlenmeyer) | ||
* Incubate à 37°C / 300 rpm and measure the OD<sub>600</sub> every 20 min | * Incubate à 37°C / 300 rpm and measure the OD<sub>600</sub> every 20 min | ||
* Determine the doubling time population and compare the strains containing or not the plasmid with a contitutive promoter and a fluorescent reporter gene | * Determine the doubling time population and compare the strains containing or not the plasmid with a contitutive promoter and a fluorescent reporter gene | ||
Latest revision as of 12:45, 21 October 2008
Culture of Stable strain with biobricks 2008
Glycerol Stocks
Minipreps (QIAGEN kit)
ElectrophoresisAn electrophoresis can be done to check if there is Product of Miniprep
Concentration of the Miniprep or the MidiprepBy biophotometry
Check if the ratio 260/280 is over 1,6 Think about the dilution ! If the concentration is to low, concentrate the sample in a vacuum centrifuge. Don't forget to open the tubes! Digestion
Migration after digestion
Separate each sample by an empty well ! Extraction
Antarctic phosphataseTo avoid autoligation, the vector in a construction has to be treated with phosphatase to dephosphorylate the cut end of the plasmid. The Antarctic phosphatase has the advantage to be easily and totally inactivated by heating.
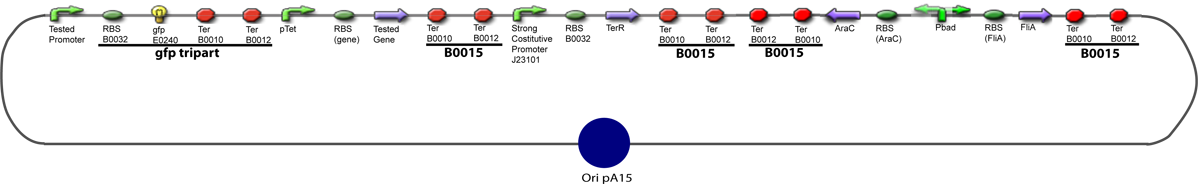
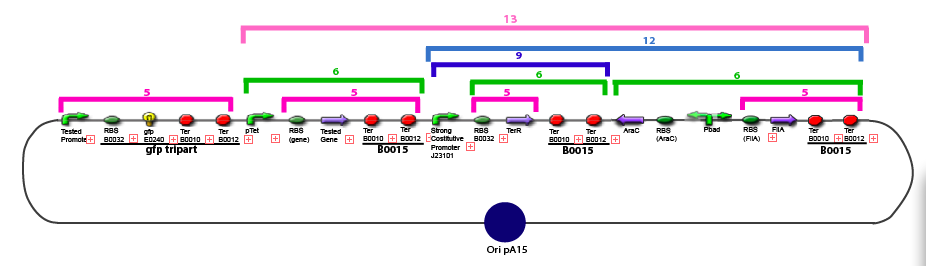
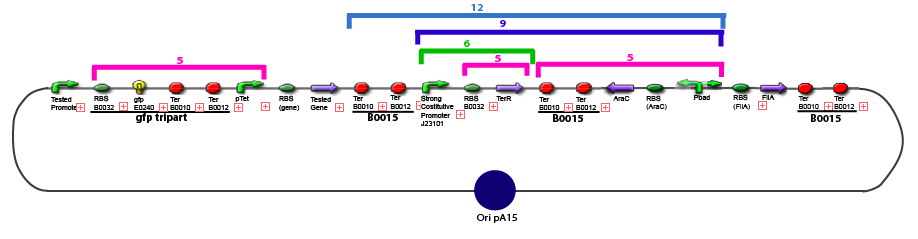
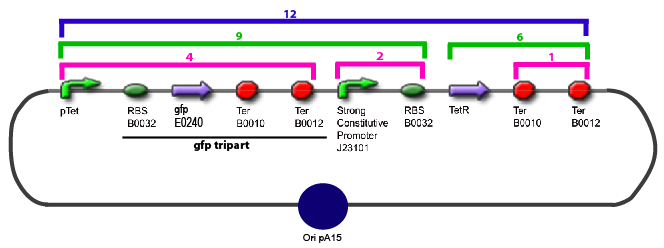
Amplification of promoters(to amplify the sequence in order to have enough amount of DNA to carry out the following of our experiments)
For each samples
1 µl dNTP
LID : 105°C Purification (PROMEGA or QIAcube or QIAquick kit)Gel Slice and PCR Product PreparationDissolving the Gel Slice
Processing PCR reactionsFor products above 40 pb
Binding of DNA
Washing
Elution
Quantification by Electrophoresis
Ligation
TransformationUse chemically lab-made competent cells or from a kit (DH5α, TOP10, Mach1)
LB and LBA
Minimum Medium
- B1 Vitamin 0.1%
Filter the medium with a cell-culture unit of filtration. If needed add the appropriate antibiotic to a final concentration of 1x Soft Agar Medium
1000x stock antibiotic
PCR ScreeningUse of 8 clones of Ligation transformants for screening PCR
After, add
Store the tubes on ice waiting for PCR attains 95°C then put the tubes in the machine
LID 105°C Electrophoresis Purification of PCR
Sequencing[http://institut.cochin.inserm.fr/rubric_recherche/Plates-Formes/sequencage_genomique/I18NFolder.2005-02-10.4781618697/page2/fr Sequencing COCHIN]
Promoter Characterization PlanFor theoretical consideration, see estimation of parameters
The same colour coded steps can be perfomed at the same time if elements needed are available The order for treating the colours should of course be:
This table contains the promoters we need to characterize, the transcription factors whose effect on the promoter we want to test, and the plasmid we want to obtain in order to carry out each characterization
Protocol to make competent bacteria1. Use non competent bacteria (ex: MG1655) stocked in 1.5 mL LB (20% Glycerol): put a sterile tip in the 1.5 mL stock tube and then place it in a 50 mL Falcon with 5 ml LB medium. Over Night culture at 37°C / 300 rpm 2. 1/100 dilution in LB medium QSP 50 mL in an erlenmeyer of 250 mL 3. Culture at 37°C / 300 rpm untill OD600 reach 0.6 4. Fast cooling at +4°C by gently shaking the erlen in ice Before: prepare CaCl2 0.1M.
5. Use pre-cooled centrifuge at +4°C. Centrifuge 50 mL of the culture in 50 mL falcon: +4°C / 5 min / 5000 rpm 6. Discard supernatant by inverting the tube, and resuspend the pellet with 1 mL of cold CaCl2 and mix gently the suspension by up and down 7. Add cold CaCl2 QSP 20 mL and incubate 30 min / +4°C 8. Centrifuge the suspension : +4°C / 5 min / 5000 rpm 9. Discard supernatant by inverting the tube, and resuspend the pellet with 1 mL of cold CaCl2 and mix gently the suspension by up and down 10. Transform or freeze the competent cells. Freeze the competent cells in 50 µL aliquots in the 0.1M CaCl2 medium with 15% glycerol. 11. After transformation, prepare a Glycerol Stock or/and use the transformed bacteria to study the doubling time of the bacteria population Study of the doubling time of the bacteria population
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"