Team:Illinois/Antibody Receptor Tyrosine Kinase Fusion Notebook
From 2008.igem.org
| Home | Team | Project | Notebook | Research Articles | Parts | Protocols | Pictures |
Contents |
July 1, 2008
Made Yeast Media
YPD Medium, per liter:
10g yeast extract
20g peptone
20g dextrose
20g agar(only for plates)
1. Dextrose filter sterilized, the rest autoclaved
2. Weigh nutrients into flask double the volume you want to make, and stir to dissolve
3. Dextrose added to autoclaved media to equivalent of 20 g/L
4. Liquid media placed on bench, plate media placed in 65 degree water bath approximately 5 minutes
5. Poured into plates and allowed to solidify
July 17, 2008
E.Coli Media
LB medium, per liter
10g tryptone
5g yeast extract
5g NaCl
1 mL 1N NaOH
15g (agar for plates)
1. Antibody DNA resuspended in TE buffer, 0.1 μg/μL
2. 5mL LB inoculated with single colony DH5a pro
3. Incubated at 37 degrees overnight
July 18, 2008
Competent cells:
1. 3mL overnight culture of DH5a pro
2. Inoculated into 35mL of LB
3. OD600 checked, want 0.2-0.3
4. Place culture on ice for 3 minutes
5. Spin at 10,000 rpm for 7 minutes, discard supernatant
6. Resuspend in 10 mL cold 30 mM CaCl2, incubated on ice overnight
July 19, 2008
Glycerol added to 10%, cells in freezer
Transformation:
1. Cells put in ice 30 min with 1μL plasmid(100ng)
2. Heat Shock for 2 minutes at 42 degrees
3. Ice for 8 minutes
4. Add cells to 1μL LB
5. Grow for 1 hour
6. Plate 100 to 200 μL on LB and amp, grow overnight
July 21, 2008
Observations:
Plenty of colonies on all plates
Also, making 5mL overnight cultures for mini-prep tomorrow.
July 22, 2008
Mini spin prep on LC and HC colonies
Place DNA in 100μL H2O in freezer
July 29, 2008
All primers brought to standard concentration, 30mM
33.3μL H2O added per n mole of primer
July 30, 2008
Assembly PCR: amplify antibody gene fragments and then assemble them, light and heavy chains
| tube | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| Antibody chain | H | H | L | L | H | H | L | L |
| DNA source | mini-prep | synthesized IDT genes | ||||||
| template | 0.5ul | |||||||
| primers | 1ul of appropriate forward and reverse primer | |||||||
| MgCl2 | 3ul | 5ul | 3ul | 5ul | 3ul | 5ul | 3ul | 5ul |
| master mix | 20ul | |||||||
| H20 | to 50ul total volume | |||||||
Master mix already contains MgCl2; should have added extra to some tubes.
PCR program: 1. 94 degrees 5 min 2. 94 degrees 1 min 3. 50 degrees 1 min 4. 72 degrees 1 min 5. GOTO 2. 39 cycles 6. HOLD at 4 degrees
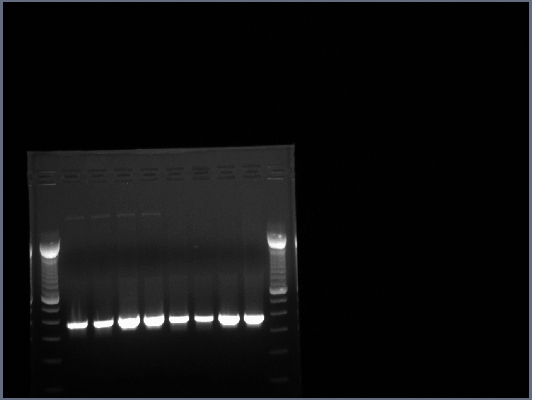
1.5% agarose gel made, 0.75g agarose in 50ml 0.5X TBE w/10ul EtBr.
20ul of PCR produts loaded on gel w/4ul 6X loading buffer, run at 200V for about half an hour.
All lanes had lots of DNA at ~375bp, PCR worked.
July 31, 2008
PCR to asemble fragments from yesterday.
| tube | 1 | 2 | 3 | 4 | ||||
| template from 7-30 PCR | 2+3 | 2+7 | 6+7 | 1+8 | ||||
| template | 0.5ul of both heavy and light chains | |||||||
| primers | 1ul of appropriate forward and reverse primer | |||||||
| MgCl2 | 0ul | 0ul | 3ul | 3ul | ||||
| master mix | 20ul | |||||||
| H20 | to 50ul total volume | |||||||
PCR program: 1. 94 degrees 5 min 2. 94 degrees 1 min 3. 50 degrees 1 min 4. 72 degrees 1 min 5. GOTO 2. 29 cycles 6. HOLD at 4 degrees
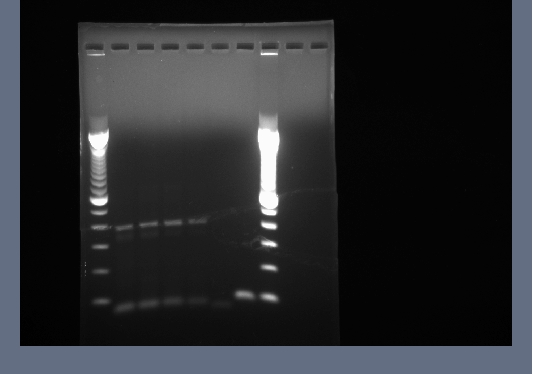
Products run on a 1.5% agarose gel, no products of 700-800bp.
August 1, 2008
PCR troubleshooting of yesterday's reaction: use gradient for annealing temp (40-65 degrees), use more template, leave out end primers
Four series of reactions using a gradient annealing temp were run in addition to a fifth series omitting the end primers.
| reaction series | A | B | C | D | E |
| template (5+7) from 7-30 PCR | 0.5ul | 1ul | 1.5ul | 2ul | 1ul |
| primers | 1ul of forward and reverse primers (omitted in E series) | ||||
| master mix | 20ul | ||||
| H20 | to 50ul total volume | ||||
annealing temps: 40, 43.8, 50, 54, 60, and 64 degrees
PCR program: 1. 94 degrees 5 min 2. 94 degrees 1 min 3. annealing step 1 min 4. 72 degrees 1 min 5. GOTO 2. 29 cycles 6. HOLD at 4 degrees
PCR products run on 1.5% agarose gel at 200V for ~20 minutes, then 240V for ~20 minutes.
Products in the 700-800bp range in all lanes. Perhaps it worked because of using a differnt product from the 7-30 PCR.
August 6, 2008
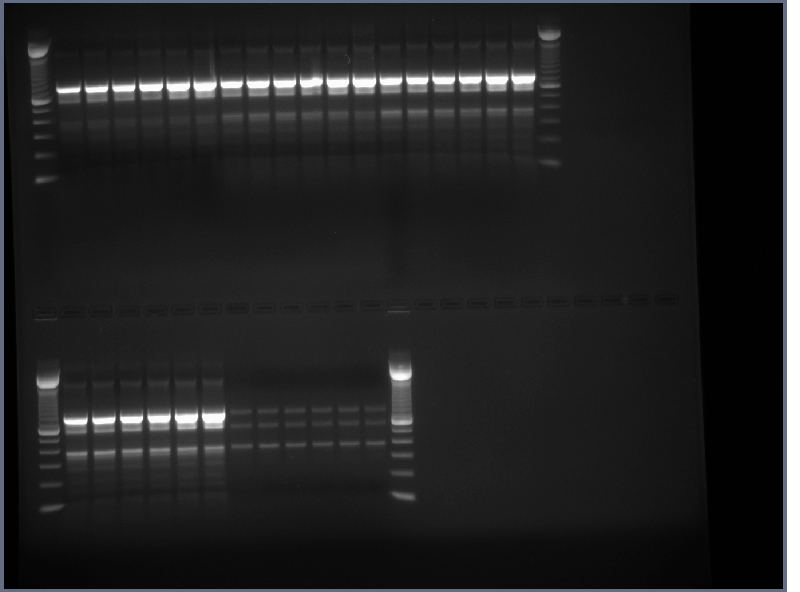
1.5% agarose gel of 8-1 PCR products run to cut out bands. Products A1, B1, C1, and E1 used.
Bands around 800bp cut out for all lanes and invitrogen gel extraction kit used to collect DNA in 50ul H2O.
September 5, 2008
Flk-1 clones from [http://www.atcc.org/ATCCAdvancedCatalogSearch/ProductDetails/tabid/452/Default.aspx?ATCCNum=MGC-18600&Template=mgcMouseClones ATCC] streaked onto LB+amp and incubated at 37 degrees.
September 6, 2008
Colonies picked from flk-1 plate and inoculated into 5ml LB+amp, grown at 37 degrees.
Freeze-dried e. coli with YCp50-poly from [http://www.atcc.org/ATCCAdvancedCatalogSearch/ProductDetails/tabid/452/Default.aspx?ATCCNum=87555&Template=vectors ATCC] resuspended in 5ml LB, grown at 37 degrees.
September 7, 2008
Mini spin prep of flk-1 and YCp50-poly cells, plasmids stored in 50ul H2O.
| Home | Team | Project | Notebook | Research Articles | Parts | Protocols | Pictures |
 "
"