Team:Cambridge/Voltage/Lab Work
From 2008.igem.org
| (7 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
| + | {{Cambridge/Notitle}} | ||
| + | <div class=bodytable> | ||
| + | {{Cambridge08}} | ||
| + | {{Cambridge08voltage}} | ||
| + | <html> | ||
| + | <table style="background:#444444; padding:15px;"> | ||
| + | <table align=left width= border="0" style="background:#444444; padding:5px;"> | ||
| + | <tr> | ||
| + | <td style="width:100%; height: 400; padding-left: 15px;"> | ||
| + | <b class="b1f"></b><b class="b2f"></b><b class="b3f"></b><b class="b4f"></b> | ||
| + | <div class="contentf"> | ||
| + | <div style="height: 400; background: white; line-height:170% padding: 5px;"> | ||
| + | <div style="color: white; font: 2px;">x</div> | ||
| + | </html> | ||
=15th July= | =15th July= | ||
==Conference in Edinburgh== | ==Conference in Edinburgh== | ||
| Line 22: | Line 36: | ||
-Designed two plasmids: | -Designed two plasmids: | ||
| - | * [[Kdp Plasmid]] | + | * [[IGEM:Cambridge/2008/Notebook/Voltage/Gene_Design#KdpF-C_Biobrick-_BBa_K090003.3D|Kdp Plasmid]] |
| - | * [[Glu Gated channel Plasmid]] | + | * [[IGEM:Cambridge/2008/Notebook/Voltage/Gene_Design#GluR0_Biobrick-_BBa_K090002|Glu Gated channel Plasmid]] |
-Sourced V.harveyi BB170 culture to obtain glutamate-gated potassium channel gene | -Sourced V.harveyi BB170 culture to obtain glutamate-gated potassium channel gene | ||
| Line 32: | Line 46: | ||
-Designed primers for extraction of Kdp Operon from E.coli and Glu Gated channel gene from V.harveyi | -Designed primers for extraction of Kdp Operon from E.coli and Glu Gated channel gene from V.harveyi | ||
| - | - | + | -Meeting with Julia Davies |
| + | |||
| + | -Discussed feasibility | ||
| + | |||
| + | -Osmotic problems with ion sequestration, possible solution - "inert" sugars such as sorbitol and manitol | ||
| + | |||
| + | -Glutamate/glutamine specificity problems, may need glutamine- as well as glutamate-free growth medium | ||
| + | |||
| + | -Need to test efficacy of voltage creation over range of osmolarities, use flame photometer to plot total K<sup>+</sup> as f<sup>n</sup> of growth curve | ||
| + | |||
| + | -Discussed Kch<sup>-</sup> mutants, generally benign | ||
| + | |||
| + | -Potential PD measurement methods: high resistance input voltmeter to minimise noise, glass capillary microelectrode, oxygen electrode | ||
| + | |||
| + | -Measurement issue of cells acting as charged particles, neutralising charge around electrode | ||
-Decided on oxygen electrode for small voltage change measurements | -Decided on oxygen electrode for small voltage change measurements | ||
| + | |||
-Extracted BioBricks from registry to use in plasmids: | -Extracted BioBricks from registry to use in plasmids: | ||
| Line 563: | Line 592: | ||
| - | |||
| - | |||
| - | |||
| - | |||
| Line 590: | Line 615: | ||
* Various combinations of restriction digests & T4 phage genome ligation test. | * Various combinations of restriction digests & T4 phage genome ligation test. | ||
* All results positive :-) | * All results positive :-) | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| Line 620: | Line 639: | ||
| - | = | + | =5th September= |
| + | ==STOP== | ||
| + | * Transform B1002 and B1006 into TOP10 | ||
| - | |||
| + | |||
| + | ==GluR0== | ||
| + | *Cut with E+P or E+S | ||
| + | *Cut pSB4C5 with same | ||
| + | *Dephosphorylate pSB4C5 | ||
| + | *Gel Extract, Ligate, Transform. | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | =6th September= | ||
| + | ==Ligations== | ||
| + | * KDP and OsmY into pSB4C5 using E+S or X+P respectively | ||
| + | * Attempt to double stop with B1006 and B1002 | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | =9th September= | ||
| + | ==Transformation== | ||
| + | * Ligations from previous day transformed into TOP10 | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | =10th September= | ||
| + | ==Analysis== | ||
| + | * Analyse transformants | ||
| + | |||
| + | ==Ligation== | ||
| + | *Re-attempt KDP/OsmY ligation | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | =11th September= | ||
| + | ==Ligation== | ||
| + | * GluR0 + pSB4T5 | ||
| + | |||
| + | |||
| + | |||
| + | =12th September= | ||
| + | ==Analysis== | ||
| + | * Analyse KDP, OsmY, Glur0 ligations | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | =15th September= | ||
| + | ==Plasmid Prep== | ||
| + | * KDP, OsmY and GluR0 | ||
| + | |||
| + | ==Restriction Digest== | ||
| + | *Digested prepped plasmids and run on gel. | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | =16th September= | ||
| + | ==Ligations== | ||
| + | * GluR0 + pSB4T5 or pSB3T5 | ||
| + | * B1002 + B1006 | ||
| + | |||
| + | ==Gel== | ||
| + | * B1002 + B1006 ligation product run with GluR0 unsupercoiled | ||
| + | ** GluR0 was lightly irradiated to induce sing strand breaks to allow it to de-supercoil and act as a 4kb template. | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | =18th September= | ||
| + | ==Applications== | ||
| + | * Designed standard infusion primers for biobrick assembly. Abandoned due to unfeasibly large scar sites between Biobricks, more effective to use custom designed primers for each reaction. | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | =19th September= | ||
| + | ==GluR0 Testing== | ||
| + | * Tested GluR0 expression cells with shiny/expensive/confusing/very, very, very old electrical equipment. Engineers left. Damn (and blast) them. The filthy varmints. | ||
| + | *Initial results promising. | ||
| + | |||
| + | |||
| + | |||
| + | =22nd September= | ||
| + | ==PCR== | ||
| + | * Single colony PCR of GluR0, OsmY, KDP. | ||
| + | * Results were ambiguous or negative. | ||
| + | |||
| + | |||
| + | |||
| + | =23rd September= | ||
| + | ==Plasmid Prep== | ||
| + | * Minipreps of some cells from the previous day | ||
| + | |||
| + | ==Gel== | ||
| + | * Plasmids from miniprep run on gel. | ||
| + | * Results negative or ambiguous. | ||
| + | |||
| + | |||
| + | |||
| + | =24th September= | ||
| + | ==Restriction Digest== | ||
| + | * GluR0 plasmid cut (E or E+P) and run on gel. | ||
| + | * Results negative | ||
| + | |||
| + | ==Plasmid Prep== | ||
| + | * KDP and some OsmY colonies. Run on gel. Best colony selected for continued work. | ||
| + | |||
| + | |||
| + | |||
| + | =25th September= | ||
| + | ==Plasmid Prep== | ||
| + | * Miniprep on best cells containing KDP, OsmY and GluR0. | ||
| + | |||
| + | ==Restriction Digest== | ||
| + | * Plasmids cut with various combinations of enzymes. | ||
| + | * Run on gel, results ambiguous. | ||
| + | |||
| + | |||
| + | =26th September= | ||
| + | ==Sequencing== | ||
| + | * Samples PCRed and cleaned, prepared for sequencing. | ||
| + | |||
| + | |||
| + | |||
| + | <!-- ## Do not edit below this line unless you know what you are doing. ## --> | ||
| + | <html> | ||
| + | <div style="color: white; font: 2px;">x</div> | ||
| + | </div></div> | ||
| + | <b class="b4f"></b><b class="b3f"></b><b class="b2f"></b><b | ||
| + | class="b1f"></b> | ||
| + | </td></tr></table></table> | ||
| + | </html> | ||
| + | __NOEDITSECTION__ | ||
__NOTOC__ | __NOTOC__ | ||
Latest revision as of 23:31, 29 October 2008
|
x
15th JulyConference in EdinburghMeet with the other UK iGEM teams.
16th JulyConference in EdinburghPresentation of projects to the other teams and to the attendees of the SynBioStandards network meeting.
17th JulyProgress-Ordered mutant E.coli chassis to test: Kch-, KdpD-, KdpE-, KdpF-E- -Designed two plasmids: -Sourced V.harveyi BB170 culture to obtain glutamate-gated potassium channel gene -Modelled protein structure -Designed primers for extraction of Kdp Operon from E.coli and Glu Gated channel gene from V.harveyi -Meeting with Julia Davies -Discussed feasibility -Osmotic problems with ion sequestration, possible solution - "inert" sugars such as sorbitol and manitol -Glutamate/glutamine specificity problems, may need glutamine- as well as glutamate-free growth medium -Need to test efficacy of voltage creation over range of osmolarities, use flame photometer to plot total K+ as fn of growth curve -Discussed Kch- mutants, generally benign -Potential PD measurement methods: high resistance input voltmeter to minimise noise, glass capillary microelectrode, oxygen electrode -Measurement issue of cells acting as charged particles, neutralising charge around electrode -Decided on oxygen electrode for small voltage change measurements
18th JulyWet WorkThere was no growth on the plates spread yesterday. There has been a few adjustments to the protocol. Biobrick Extraction
Bigger, better, faster, strongerWe found we could not extract enough DNA from the registry, so we are upgrading the extraction to the "bigger, better, faster, stronger" method. Warm 50μL of EB in Eppendorf tubes at 50°C and add 4 punched spots. Keep it warming at 50°C for 20mins and spin down for 3 minutes at 15,000 g. Warm agin for 10mins and spin down again for 3mins. Pipette off the liquid which should have the DNA in. We then confirmed with PCR.
5μL of DNA in EB buffer 2.5μL of each primer for Biobrick vectors 25μL of Finnzymes mastermix 15μL of sterile distilled water And all 50μL were then run through the PCR reaction and 17μL of product run on an E-Gel with 3μL dye. The results gave large amounts of DNA in each lane for all Biobricks except F. The original extracts of G,H,I,J,K and L were then used for the following transformations. To chilled tubes add 5μL of DNA + EB solution to 5μL of Cells ( Chemically competent Top 10 )
Ice for 30 minutes Heat shock at 42°C for 60 seconds Ice for 2 minutes Add 500μL of SOC Incubate at 37°C overnight
Prepare Agar plates : Add 200μL of Amp (100mg/mL) into 200mL of LA ( Final conc. of 100μg/mL ) and add 100μL of Amp (100mg/mL) into 200mL of LA ( Final conc. of 50μg/mL ) Plate neat samples of G,H,I,J,K,L onto both types of plate and incubate overnight.
21st JulyWet WorkAims for TodayThe results of yesterday's modified protocol still yielded no growth on the plates. Using yesterday's PCR product our aims were:
Length
22nd JulyWet Work
16 LA plates were poured with 8 having 25μg/mL Kanamycin, ready for mutant strains to be grown on. The gel extracts from the ligation of primer and rbs were extracted using Zymoclean gel extraction kit. Hopefully these now contain linear DNA of BBa_J23113 and BBa_B0030, and BBa_J23114 and BBa_B0030. BioBricks J45996, J45995 and J45250 were also extracted from the registry, using the "bigger, better, faster, stronger, method", in order to provide template to extracted the OsmY promoter.
23rd JulyWet Work
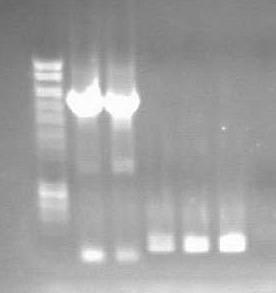
24th JulyDouble TerminatorThe double terminator was made from BBa_B1006 and BBa_B1002, combined together. First the two Biobricks were cut using the fast digest enzymes, at either Xba or Spe. They were then run on an agarose gel, cut out and extracted, using Zymoclean gel extraction. The 2006 was not cut succesfully with Spe, so was not extracted. The samples were then ligated, using fast ligation, such that 1002-1002 and 1002-1006 were hopefully produced. This was done by combining the 1002 cut with Spe with the 1002 cut with Xba and the 1006 cut with Xba. These were then run on an agarose gel and cut out, then frozen overnight.
25th JulyDouble TerminatorThe DNA was extracted from lanes 3&4 of the gel and extracted using the Zyppy Gel Extraction kit. The extracted DNA was then PCRed using 34 cycles, and then 10μL was frozen at -20°C. 8μL of the PCR product was cut with both EcoR1 and Pst1, as was 5μL of the death plasmid, PSB4C5, using the fast digest protocol. The gene was then ligated into the plasmid backbone, using the fast ligation kit, from Finnzymes. 500μL of normal TOP10 cells were made competent, and then transformed with the 4μL of the new plasmid, and grown on chloramphenicol overnight. 1μL of Puc19 was used as a control. K+ assay for E.coliSolutions of potassium chlotide were made up from 0mM to 500mM. 100μL of cells, 100μL of 10x KCl solution and 800μL of SDW were mixed and the OD600 measured every 30 mins, after agitation to ensure homogenaity. After 4 hours 800μL of the cell solution were spun down and resuspended in 1mL SDW, lysed using the freeze-thaw method, and the K+ concentration measured using the Flame Photometer.
28th JulyThe flame photometer was also calibrated using μM concentrations of potassium.
Mutant Plates
29th July
30th JulyCompetence test
osmY and Kdp
31st JulyMutants
Kdp
OsmY
1st AugustMutants
osmY
Plasmid
4th AugustBiobricks
5th AugustKdp?
Biobricks
6th AugustKdp?
Mutants
7th AugustFlame Photometry
Kdp
Mutants8th AugustKdp?
Mutants
11th AugustKdp?
OsmY and RBS
12th AugustKdp
OsmY
Make competent stocksChemically competent TOP10 cells were made by growing overnight in 200ml LB in a shaking incubater at 37°C. 200ml were spun down at 3800rpm for 8 mins, resuspended in 20ml CaCl2, spun down again and resuspended in 4ml CaCl2. Again they were spun down (6500 rpm for 5 mins)and resuspended in 4ml 60%glycerol, spun down and resuspended in 2ml 60%glycerol. Finally they were aliquoted (100μL) into stock tubes and flash frozen in liquid nitrogen. Electroporation competent TOP10 and DH5α cells were made by growing overnight in 200ml LB in a shaking incubater at 37°C. 100ml were spun down at 3800rpm for 8 mins, resuspended in 25ml SDW, spun down again and resuspended in 2ml SDW. Again they were spun down (6500 rpm for 5 mins)and resuspended in 2ml 60%glycerol, spun down and resuspended in 2ml 60%glycerol. Finally they were aliquoted (100μL) into stock tubes and flash frozen in liquid nitrogen. However lost half of the DH5α when centrifuge tube broke.
13th AugustKdp
Recovering frozen cellsTo recover frozen cells they were thawed on ice and spun down at6500rpm for 5mins. They were then resuspended in 100μL CaCl2. They were then transformed with 2μL pUC9 and grown on 100μg/mL Amp plates.
14th AugustKdp
OsmY and RBS
15th AugustKdp
MEGA Mutant Assay
16th AugustBank Holiday. Apparently no-one was quite that dedicated to come into work.
19th AugustLigation Diagnostic
Kdp
20th AugustpSB4C5 & RBS
Kdp
21st AugustOsmY RBS
22nd AugustKdp & OsmY-RBS
26th August=Kdp screening
27th AugustOsmY RBS
Restriction Digest & Ligation Test
29th AugustGluR0
2nd SeptemberWE HAVE KDP!
5th SeptemberSTOP
GluR0
6th SeptemberLigations
9th SeptemberTransformation
10th SeptemberAnalysis
Ligation
11th SeptemberLigation
12th SeptemberAnalysis
15th SeptemberPlasmid Prep
Restriction Digest
16th SeptemberLigations
Gel
18th SeptemberApplications
19th SeptemberGluR0 Testing
22nd SeptemberPCR
23rd SeptemberPlasmid Prep
Gel
24th SeptemberRestriction Digest
Plasmid Prep
25th SeptemberPlasmid Prep
Restriction Digest
26th SeptemberSequencing
x
|
 "
"