Team:Hawaii/Notebook/2008-08-31
From 2008.igem.org
| Projects | Events | Resources | ||
|---|---|---|---|---|
| Sponsors | Experiments | Milestones | Protocols | |
| Notebook (t) | Meetings (t) |
Things we did today
Wetlab work
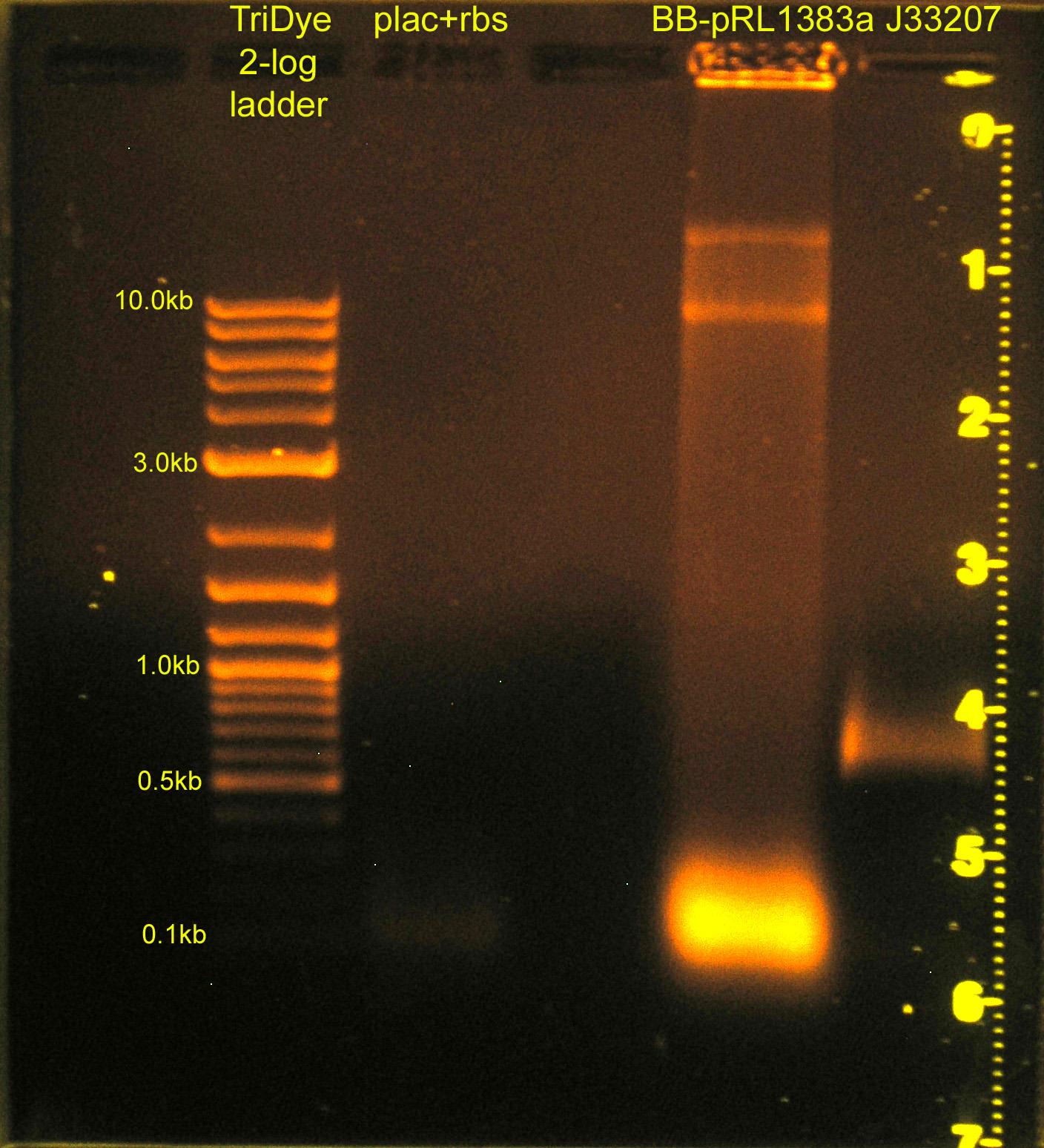
Construction of p+r (cont.) and re-replacement of BB-pRL1833a MCS
- Grace
- Ran RE digests from last night on gel
- plac+rbs band not visible; will ligate using plac and rbs parts (instead of plac+rbs part)
- Image to right has been edited w/ Photoshop. Faint smear for plac+rbs was not visible before.
- Extracted bands from gel
- Ligated:
- plac and rbs (B0034) and C0012 vector
- J33207 and BB-pRL1383a
- Transformed into DH5α cells
- Plated BB-pRL1383a+J33207 on selective media with and without IPTG to verify if IPTG is necessary for blue/white screening in this strain
Inoculated TB+sp100 with BB-pRL1383a
- Grace
Construction of Broad-Host-Range Plasmid Parts
- Margaret
File:Re digest 8 31 08.jpg
Restriction digest of rep and P1 lytic regions.
- gel of yesterday's re-digest, lane 3 was cleaned using gel purification spin columns and stored in -20°C. Lanes 7-10 were extracted and cleaned as well.
- Ligation: using T4 ligase, 10X buffer (from the same lot as the enzyme, it was aliquoted today). 2 hour incubation at room temperature. The reaction was placed in 4°C in case transformation does not yield colonies.
| insert | vector |
|---|---|
| 1 pSB1A3 cut with X,P(de-P042-)18ng | 3 rep (PCR product) X,P 82ng |
| 1pSB1A3 cut with X,P(de-P042-)35.6ng | 3P1 lytic region X,P 64.4ng |
| 3pSB1A3 cut with X,P(de-P042-)66.2ng | 1 rep (PCR product) X,P 33.8ng\ |
- Transformation: of ligation products from today into DH5-alpha cells. Everything plated on Amp100 LB plates, pSMC121 used as positive control (plated on SmSp).
- Plasmid Prep: stored in -20°C in 30ul TE buffer
- Culture: 5mL Terrific Broth + amp100
- plac+B0030 (1:6):3, 4, 7
- plac+B0030 :3
- plac+B0034 (1:6):1
- plac+B0034 (1:3):3
Discussion
Quote of the Day
History is the only laboratory we have in which to test the consequences of thought. - Étienne Gilson
 "
"