Team:Heidelberg/Notebook/Sensing Group/Notebook/6thweek
From 2008.igem.org


Contents |
Monday, 09/08/2008
- Culture of 5 colonies from LuxQ Transformation and 1 from F1 transformation
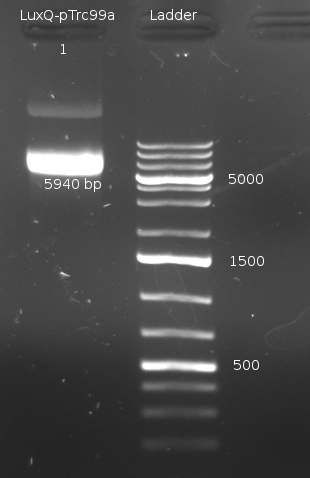
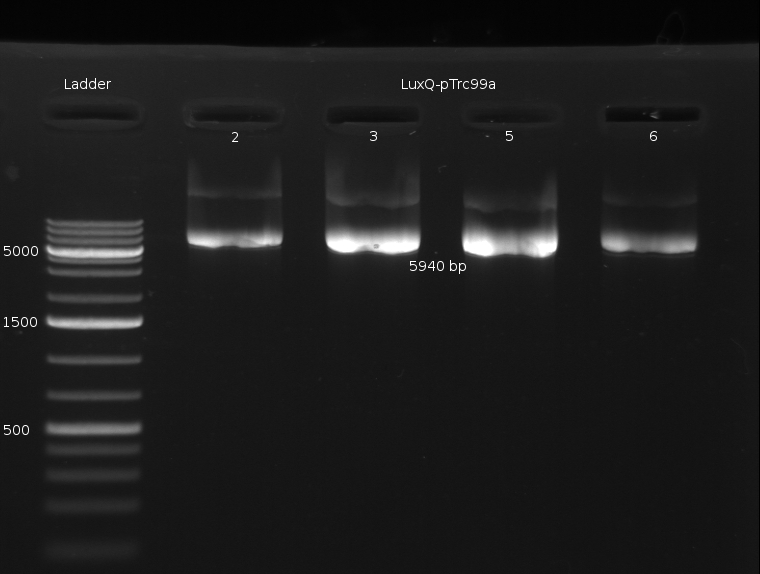
- Miniprep of Culture from previous Transformation (done by Chenchen). Incubated for two days @ RT and about then 6 h @ 37 °C
- Digestion of the Miniprep products with XbaI (NEBuffer 2 + BSA, 1 ul enzyme @ 37 °C about 40min)
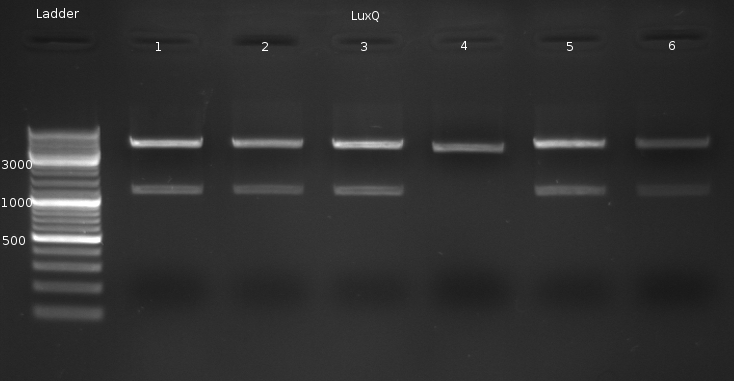
Sequencing @ GATC verified correct sequence for LuxQ no. 1
- PCR for LuxQ and LuxP with Phusion (25 µl Mastermix, 0.5 µl each Primer, 0(1) µl DMSO, 24(23) µl H20, 0.5 µl Template)
- PCR Programme: 5 min @ 98 °C || 10 s @ 98 °C | 30 s @ 55 °C | 30 s @ 72 °C || 5 min @ 72 °C | 4 °C
Tuesdsay, 09/09/2008
- Miniprep of LuxQ colonies no. 1,2,3,5,6
- PCR for LuxQ fragment 1 and 2, Tar fragment 1 and 2 for Phusion receptor with Phusion-Polymerase
- PCR Programme: 30 s @ 98 °C || 10 s @ 98 °C | 30 s @ 55 °C |30 s @ 72 °C || 5 min @ 72 °C | 4 °C (30 cycles)
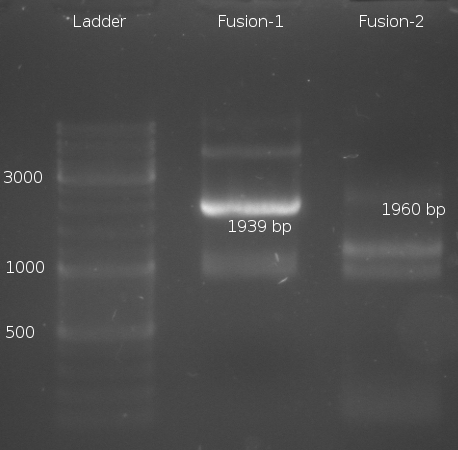
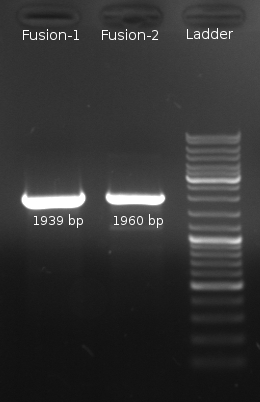
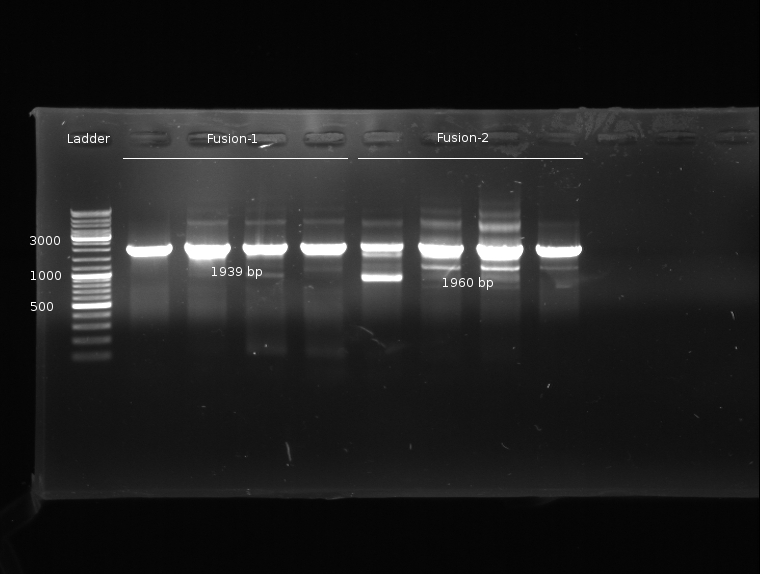
- Fusion-PCR for Fusion-1 and Fusion-2 constructs
- O/N Culture of 10 LuxP in pDK48 colonies
Wednesday, 09/10/2008
LuxP cloning
- Miniprep of O/N LuxP cultures and digestion with NcoI (NEBuffer 2, 2h @ 37 °C)
Sequencing @ GATC: LuxP1 correct
Fusion chimeras
- PCR for LuxQ 1+2, Tar 1+2 and Gel Extraction to get rid of first primers
- Fusion-PCR of LuxQ1+Tar1 und LuxQ2+Tar2 with Phusion and 55 °C annealing --> No product
- Fusion PCR of LuxQ fragment 1b, LuxQ fragment 1c, LuxQ fragment 2b, LuxQ fragment 2c, Tar fragment 1b, Tar fragment 1c, Tar fragment 2b, Tar fragment 2c with Phusion and 55 °C annealing. Products were purified via Gelextraction to get rid of first primers
- Two Fusion PCR (30 s @ 98 °C || 10 s @ 98 °C | 30 s @ 55 °C |30 s @ 72 °C || 5 min @ 72 °C | 4 °C (30 cycles)) with PCR purification.
- Gel Extraction of LuxQ 1+2, Tar 1+2
Thursday, 09/11/2008
Fusion chimeras
- PCR Purification of previous Fusion PCR
- Digestion with NcoI/NdeI (NEBuffer4) and NcoI/KpnI (NEBuffer1 + BSA). 1 h @ 37 °C
- Gel extraction, eluted in 30 µl water
- Ligation (Insert:Vector 5 µl:5 µl) and transformation into DH5alpha competent cells
Friday, 09/12/2008
nothing to report
 "
"