Team:Paris/August 27
From 2008.igem.org
(Difference between revisions)
(→Digestion) |
(→Electrophoresis) |
||
| Line 165: | Line 165: | ||
No correct clone | No correct clone | ||
<br>The 8 other clones were also screened. | <br>The 8 other clones were also screened. | ||
| - | + | [[Image:KR000256.JPG|thumb|]] | |
'''PCR''' | '''PCR''' | ||
elongation time: 2 min 30 | elongation time: 2 min 30 | ||
| Line 229: | Line 229: | ||
|} | |} | ||
<br> | <br> | ||
| - | |||
=Cloning of OmpR*= | =Cloning of OmpR*= | ||
Revision as of 18:49, 27 August 2008
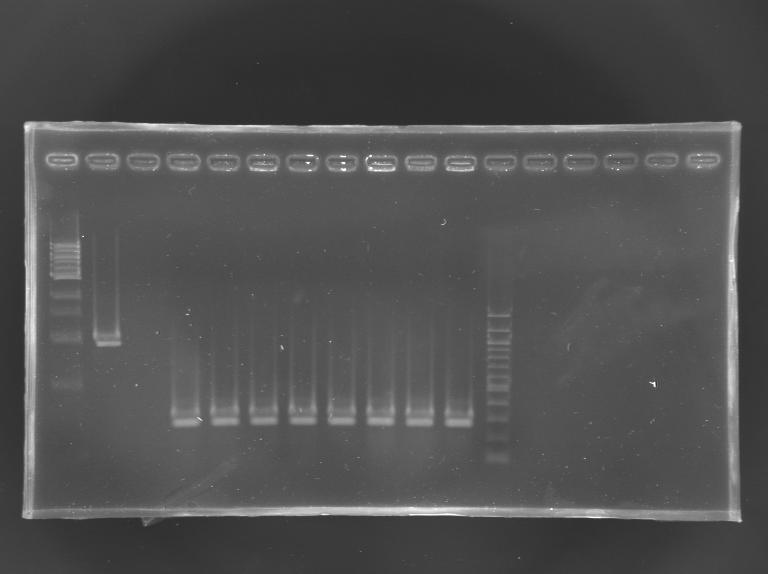
Construction of pFlhB - mRFP Tripart (LVA+)Aim : Construction of "pFlhB-RBS-mRFP-dbl ter" (pFlhB-I732078) DigestionMeasurement of the concentration of D187 purifiedProtocol (it's same that for Miniprep) => the experiments of Gel Extraction have failed, so we need to repeat the step of digestion. Digestion
Gel VerificationFile:KR000.JPG Gel Verification of D187 digestion
Cloning of EnvZ* in pSB1A2Transformation results
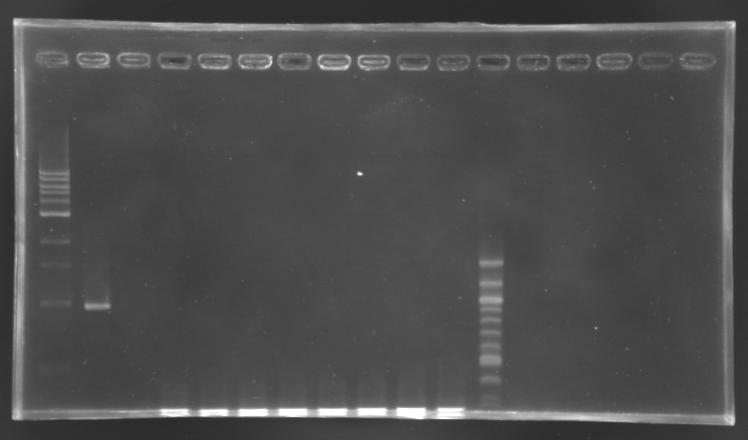
PCR screening
Electrophoresis
PCR
elongation time: 2 min 30
Cloning of OmpR*DigestionDetermination of the concentration of DNAWe used the biophotometer
Name of the digestions
Protocol of digestion
Cleaning of the digestion productsLigationDetermination of the concentration of DNAWe used the biophotometer
Protocol of ligation L171
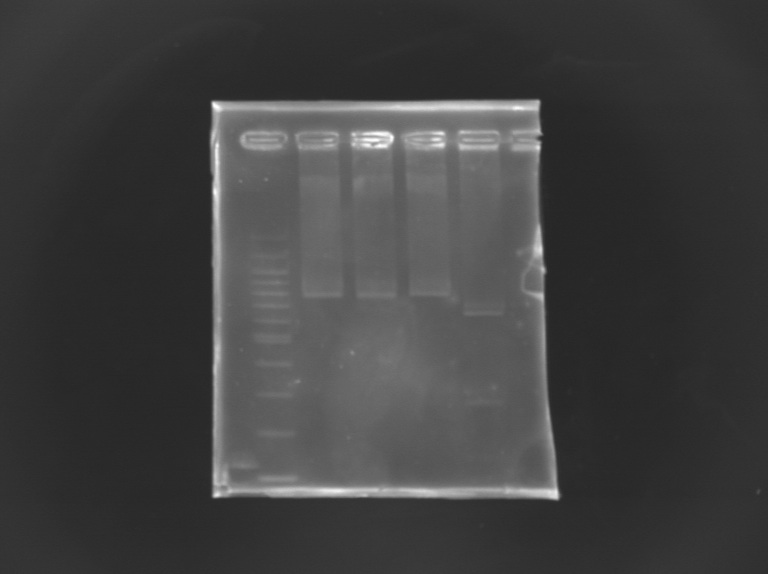
Checking mutagenesis FliAFor this, i digested mutated FliA and non-mutated FliA with EcoRI and PstI and put in migration the digestion products running on gels. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"