Team:Heidelberg/Notebook/Sensing Group/Notebook/2ndweek
From 2008.igem.org


Contents |
Monday, 08/11/2008
- preparation of LuxS harboring cells O/N culture
Tuesday, 08/12/2008
- Miniprep of correctly sequenced LuxS and Glycerol-stock
- LuxQ no. 3 checked via digestion with NcoI/BamHI (NEBuffer 3 + BSA) --> negative result
- PCR for LuxQ, LuxQ-1, LuxQ-2, Tar-1, Tar-2 with Taq Mastermix
- 5min @ 95°C || 30s @ 95°C | 30s @ 58 °C | 2min @ 72°C || 10min @ 72°C | 4°C hold (30 cycles)
- preparation of O/N culture for LuxQ no. 3
Wednesday, 08/13/2008
- PCR-purification of the all PCR-products eluted in 50 µl H2O
- Miniprep of LuxQ no. 3 O/N culture and digestion with xbaI (NEBuffer 2 + BSA)
- Fusion-PCR for Fusion-1 and repetition of PCR for LuxQ-2 and LuxQ
- digestion of pDK48 plasmid and Fusion-1 frament with NcoI/NdeI (0.5 µl) and AgeI (1 µl) in NEBuffer 4
Thursday, 08/14/2008
- Gel for LuxQ, LuxQ2
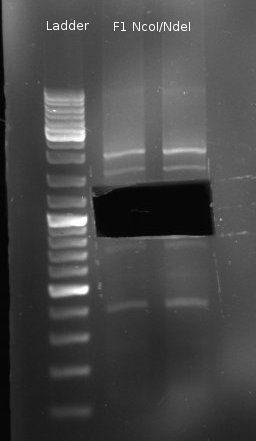
- sequentiell digestion of Fusion-1 with NcoI and NdeI (NEBuffer 3)
- Double digestion of pDK48 with NcoI/NdeI (NEBuffer 4)
Friday, 08/15/2008
- digestion of Fusion-1 with AgeI in NEBuffer 1
- gel extraction of sequentially digested Fusion-1 and pDK48 products
- new digestion of Fusion-1 with NcoI/NdeI (NEBuffer 4) and gel extraction, because previous digestion did not yield expected bands (there should be two small bands at 516bp and 392bp)
- Ligation of 5 µl Fusion-1 with 5 µl pDK48 (both digested with NcoI/NdeI )
- Transformation into DH5α (5 µl Ligation products)
 "
"