Team:UNIPV-Pavia/Notebook/Week8
From 2008.igem.org
Notebook
| Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | Week 6 | Week 7 |
|---|---|---|---|---|---|---|
| Week 8 | Week 9 | Week 10 | Week 11 | Week 12 | Week 13 | Week 14 |
Week 8: 07/7/08 - 07/12/08
07/7/08
- Colony PCR (5 colonies for each plate) for:
- BBa_J23100-BBa_B0030-BBa_C0012 (for the second time, we hoped to find true positive colonies)
- BBa_J23100-BBa_B0030-BBa_I15010 (for the second time, we hoped to find true positive colonies)
- BBa_B0030-BBa_E0040-BBa_B1006
- BBa_B0030-BBa_C0051-BBa_B0030
- BBa_B0030-BBa_E1010-BBa_B1006
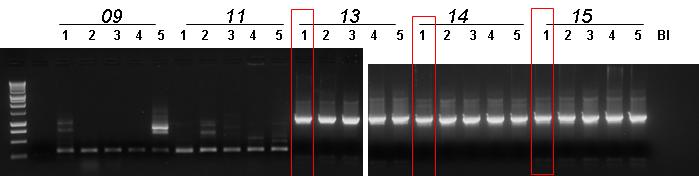
- Gel results:
- No true positives for BBa_J23100-BBa_B0030-BBa_C0012
- No true positives for BBa_J23100-BBa_B0030-BBa_I15010
- Non pure true positives for BBa_B0030-BBa_E0040-BBa_B1006
- Pure true positives for BBa_B0030-BBa_C0051-BBa_B0030
- Non pure true positives for BBa_B0030-BBa_E1010-BBa_B1006
- We chose to keep the first colony for all the 3 working ligation plates.
- NOTE: BBa_B0030-BBa_E0040-BBa_B1006 and BBa_B0030-BBa_E1010-BBa_B1006 are final parts; we decided to sequence these 2 parts even if gel showed a weak contamination: sequencing results will tell us if there are false positive plasmids in those colonies or if the contamination was only a PCR contamination.
- We infected 9 ml LB + suitable antibiotic with 30 µl of these glycerol stocks:
| BBa_B0030 | BBa_I15010 | BBa_B0030-BBa_E0040-BBa_B1006 (1) | BBa_B0030-BBa_E1010-BBa_B1006 (1) |
| BBa_C0012 | BBa_R0062 | BBa_B0030-BBa_C0051-BBa_B0030 | BBa_J23100-BBa_B0030 |
- We incubated the 8 cultures at 37°C, 220 rpm overnight.
- Tomorrow we will be ready to perform NINE LIGATIONS...(@_@!)
07/8/08
- We received sequencing results for BBa_B0030-BBa_C0078: sequence showed an extra DNA fragment between BBa_C0078 and Suffix...We decided to ignore it because there was a stop codon before that fragment.
- Glycerol stocks for:
| BBa_B0030 | BBa_I15010 | BBa_B0030-BBa_E0040-BBa_B1006 (1) |
| BBa_C0012 | BBa_R0062 | BBa_B0030-BBa_C0051-BBa_B0030 |
| BBa_J23100-BBa_B0030 | BBa_B0030-BBa_E1010-BBa_B1006 (1) |
- Miniprep for these parts.
- Plasmid digestion for:
| BBa_B0030 (S-P) | BBa_B0030-BBa_E0040-BBa_B1006 (1) (E-X) | BBa_B0030-BBa_E1010-BBa_B1006 (1) (E-X) |
| BBa_C0012 (X-P) | BBa_B0030-BBa_C0051-BBa_B0030 (S-P) | BBa_J23100-BBa_B0030 (E-S) |
| BBa_I15010 (X-P) | BBa_J23100-BBa_B0030-BBa_C0040 (E-S) | BBa_R0051-BBa_B0030-BBa_C0062 (E-S) |
| BBa_R0062 (E-X) | BBa_B0030-BBa_C0061-BBa_B1006 (E-S) |
- Gel run/cut/gel extraction for:
| BBa_I15010 (X-P) | BBa_J23100-BBa_B0030 (E-S) | BBa_J23100-BBa_B0030-BBa_C0040 (E-S) |
| BBa_C0012 (X-P) | BBa_R0051-BBa_B0030-BBa_C0062 (E-S) | BBa_B0030-BBa_C0061-BBa_B1006 (E-S) |
- DNA precipitation with sodium acetate for:
| BBa_B0030 (S-P) | BBa_B0030-BBa_E0040-BBa_B1006 (1) (E-X) | BBa_B0030-BBa_E1010-BBa_B1006 (1) (E-X) |
| BBa_R0062 (E-X) | BBa_B0030-BBa_C0051-BBa_B0030 (S-P) |
- (We already had BBa_I15009(X-P) and BBa_B1006(E-X))
- Ligations:
- BBa_B0030-BBa_I15009
- BBa_J23100-BBa_B0030-BBa_C0040-BBa_B1006
- BBa_R0051-BBa_B0030-BBa_C0062-BBa_B1006
- BBa_B0030-BBa_C0051-BBa_B0030-BBa_C0079
- BBa_B0030-BBa_C0061-BBa_B1006-BBa_R0062
- BBa_J23100-BBa_B0030-BBa_C0012 (again)
- BBa_J23100-BBa_B0030-BBa_I15010 (again)
- BBa_J23100-BBa_B0030-BBa_E0040-BBa_B1006 (to test the part)
- BBa_J23100-BBa_B0030-BBa_E1010-BBa_B1006 (to test the part)
- We incubated ligations at 16°C overnight.
- We infected 9 ml LB + Amp with 30 µl of BBa_B1006 and BBa_B0030-BBa_C0078 glycerol stocks. We incubated the 2 cultures at 37°C, 220 rpm ovenight.
07/9/08
- We transformed the 9 ligations (2 µl) and plated transformed bacteria. We incubated plates at 37°C ovenight.
- Glycerol stocks for BBa_B1006 and BBa_B0030-BBa_C0078.
- Miniprep for BBa_B1006 and BBa_B0030-BBa_C0078.
- Plasmid digestion:
| BBa_B1006 (E-X) | BBa_B0030-BBa_C0078 (E-S) |
- Gel run/cut/gel extraction for BBa_B0030-BBa_C0078 (E-S).
- DNA precipitation with sodium acetate for BBa_B1006 (E-X).
- Ligation: BBa_B0030-BBa_C0078-BBa_B1006
- We incubated ligation at 16°C overnight.
07/10/08
- We transformed BBa_B0030-BBa_C0078-BBa_B1006 ligation (2 µl) and plated transformed bacteria. We incubated the plate at 37°C ovenight.
- All the ligation plates showed colonies! There were carpets, but we could pick up some single colonies for colony PCR.
- Colony PCR for:
- BBa_B0030-BBa_I15009
- BBa_J23100-BBa_B0030-BBa_C0040-BBa_B1006
- BBa_R0051-BBa_B0030-BBa_C0062-BBa_B1006
- BBa_B0030-BBa_C0051-BBa_B0030-BBa_C0079
- BBa_B0030-BBa_C0061-BBa_B1006-BBa_R0062
- BBa_J23100-BBa_B0030-BBa_C0012
- BBa_J23100-BBa_B0030-BBa_I15010
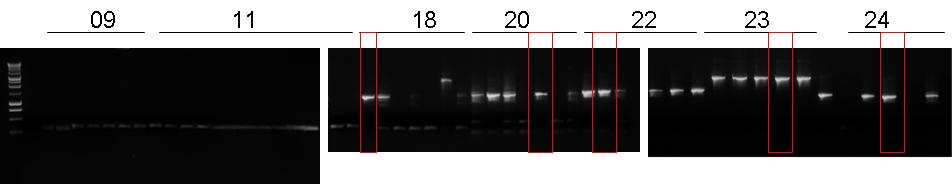
- Gel results:
- BBa_B0030-BBa_I15009 1st colony was chosen.
- BBa_J23100-BBa_B0030-BBa_C0040-BBa_B1006 5th colony was chosen.
- BBa_R0051-BBa_B0030-BBa_C0062-BBa_B1006 2nd colony was chosen.
- BBa_B0030-BBa_C0051-BBa_B0030-BBa_C0079 4th colony was chosen.
- BBa_B0030-BBa_C0061-BBa_B1006-BBa_R0062 3rd colont was chosen.
- BBa_J23100-BBa_B0030-BBa_C0012 did not show true positive colonies.
- BBa_J23100-BBa_B0030-BBa_I15010 did not show true positive colonies.
- We streaked BBa_J23100-BBa_B0030-BBa_E0040-BBa_B1006 and BBa_J23100-BBa_B0030-BBa_E1010-BBa_B1006 with a top to test the parts (NOTE: we could see some red colonies on BBa_J23100-BBa_B0030-BBa_E1010-BBa_B1006 plate!these colonies correspond to bacteria with correctly ligated plasmid, while normal color colonies correspond to bacteria with BBa_B0030-BBa_E1010-BBa_B1006 plasmid, without constitutive promoter).
- We infected 100 µl LB + Amp with the tips and incubated the culture at 37°C, 220 rpm for 2 hours.
- Then we watched green, blue and red fluorescence channels at microscope (50 µl of each culture):
- LB preparation: 0.5 l LB + Amp for plates.
- BBa_R0010 resuspension from Registry of Standard Parts.
- We transformed (4 µl) BBa_R0010 and plated transformed bacteria. We incubated BBa_R0010 plate at 37°C overnight.
- We infected 9 ml LB + suitable antibiotic with 30 µl of these glycerol stocks:
| BBa_C0012 | BBa_B0030-BBa_I15009 (1) |
| BBa_J23100-BBa_B0030-BBa_C0040-BBa_B1006 (5) | BBa_R0051-BBa_B0030-BBa_C0062-BBa_B1006 (2) |
| BBa_J23100-BBa_B0030 | BBa_B0030-BBa_C0051-BBa_B0030-BBa_C0079 (4) |
| BBa_B0030-BBa_C0061-BBa_B1006-BBa_R0062 (3) |
- We also picked up one colony from BBa_I15010 plate with a tip and infected 9 ml LB + Kan. We incubated the 8 cultures at 37°C, 220 rpm overnight.
07/11/08
- BBa_R0010 plate showed 2 colonies. We picked up one colony and infected 9 ml LB + Amp to grow an overnight culture.
- BBa_B0030-BBa_C0078-BBa_B1006 plate showed many colonies. We used 7 colonies to perform colony PCR.
- Gel results: all the 7 colonies contained ligated plasmid; we chose the 5th colony to grow a 9 ml overnigth culture because 5th it showed no impurities.
- Glycerol stocks for:
| BBa_C0012 | BBa_B0030-BBa_I15009 (1) | BBa_J23100-BBa_B0030-BBa_C0040-BBa_B1006 (5) |
| BBa_I15010 | BBa_J23100-BBa_B0030 | BBa_R0051-BBa_B0030-BBa_C0062-BBa_B1006 (2) |
| BBa_B0030-BBa_C0051-BBa_B0030-BBa_C0079 (4) | BBa_B0030-BBa_C0061-BBa_B1006-BBa_R0062 (3) |
- Miniprep for these 8 parts.
- We sent BBa_I15010 and BBa_B0030-BBa_C0061-BBa_B1006-BBa_R0062 (3) to Primm for sequencing.
- Plasmid digestion for:
| BBa_C0012 (X-P) | BBa_B0030-BBa_I15009 (1) (E-S) | BBa_B1006 (we already had this plasmid at -20°C) (E-P) |
| BBa_J23100-BBa_B0030 (E-S) | BBa_J23100-BBa_B0030-BBa_C0040-BBa_B1006 (5) (E-S) | BBa_R0051-BBa_B0030-BBa_C0062-BBa_B1006 (2) (E-S) |
| BBa_J23100-BBa_B0030 (S-P) | BBa_R0040 (we already had this plasmid at -20°C) (E-X) | BBa_B0030-BBa_C0051-BBa_B0030-BBa_C0079 (4) (E-S) |
- Gel run/cut/gel extraction for all these parts.
- (We already had BBa_B1006 (E-X) and BBa_R0062 (E-X))
- Ligation:
- BBa_J23100-BBa_B0030 (S-P) -BBa_C0012 (X-P) (1:2 ratio instead of 1:6)
- BBa_B1006 (E-P) + BBa_J23100-BBa_B0030 (E-S, 1:4) -BBa_C0012 (X-P, 1:2) (we decided to try this double ligation)
- BBa_J23100-BBa_B0030-BBa_C0040-BBa_B1006-BBa_R0040
- BBa_R0051-BBa_B0030-BBa_C0062-BBa_B1006-BBa_R0062
- BBa_B0030-BBa_C0051-BBa_B0030-BBa_C0079-BBa_B1006
- BBa_B0030-BBa_I15009-BBa_B1006
- We incubated ligations at 16°C overnight.
- We infected 9 ml LB + Amp with 30 µl of BBa_R0079 glycerol stock.
07/12/08
- Glycerol stocks for:
| BBa_R0010 | BBa_R0079 | BBa_B0030-BBa_C0078-BBa_B1006 |
- Miniprep for these 3 parts.
- We put these 3 purified plasmids at -20°C.
- We also put the 6 ovenight ligations at -20°C. Next week they will be transformed!
 "
"