Team:UNIPV-Pavia/Notebook/Week20
From 2008.igem.org
Notebook
| Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | Week 6 | Week 7 |
|---|---|---|---|---|---|---|
| Week 8 | Week 9 | Week 10 | Week 11 | Week 12 | Week 13 | Week 14 |
| Week 15 | Week 16 | Week 17 | Week 18 | Week 19 | Week 20 | Week 21 |
| Week 22 | Week 23 | Week 24 |
Week 20: 09/29/08 - 10/3/08
09/29/08
- Ligations:
- Lig.34-Lig.15 (=TR)(our RFP protein generator downstream, to check Lig.34 functionality)
- T5-Lig.15 (=TT)(our RFP protein generator after our GFP protein generator, to check terminator efficiency)
- We incubated ligations at 16°C overnight.
09/30/08
- We transformed the two overnight ligations and plated transformed bacteria. We incubated the two plates at 37°C overnight.
10/1/08
- We received sequencing results for Lig.31 and Lig.36. Sequences were OK!!! Notice that both of the two parts are long. So, sequencing could only confirm partially the nucleotides composing these two parts.
- Plates were ok!
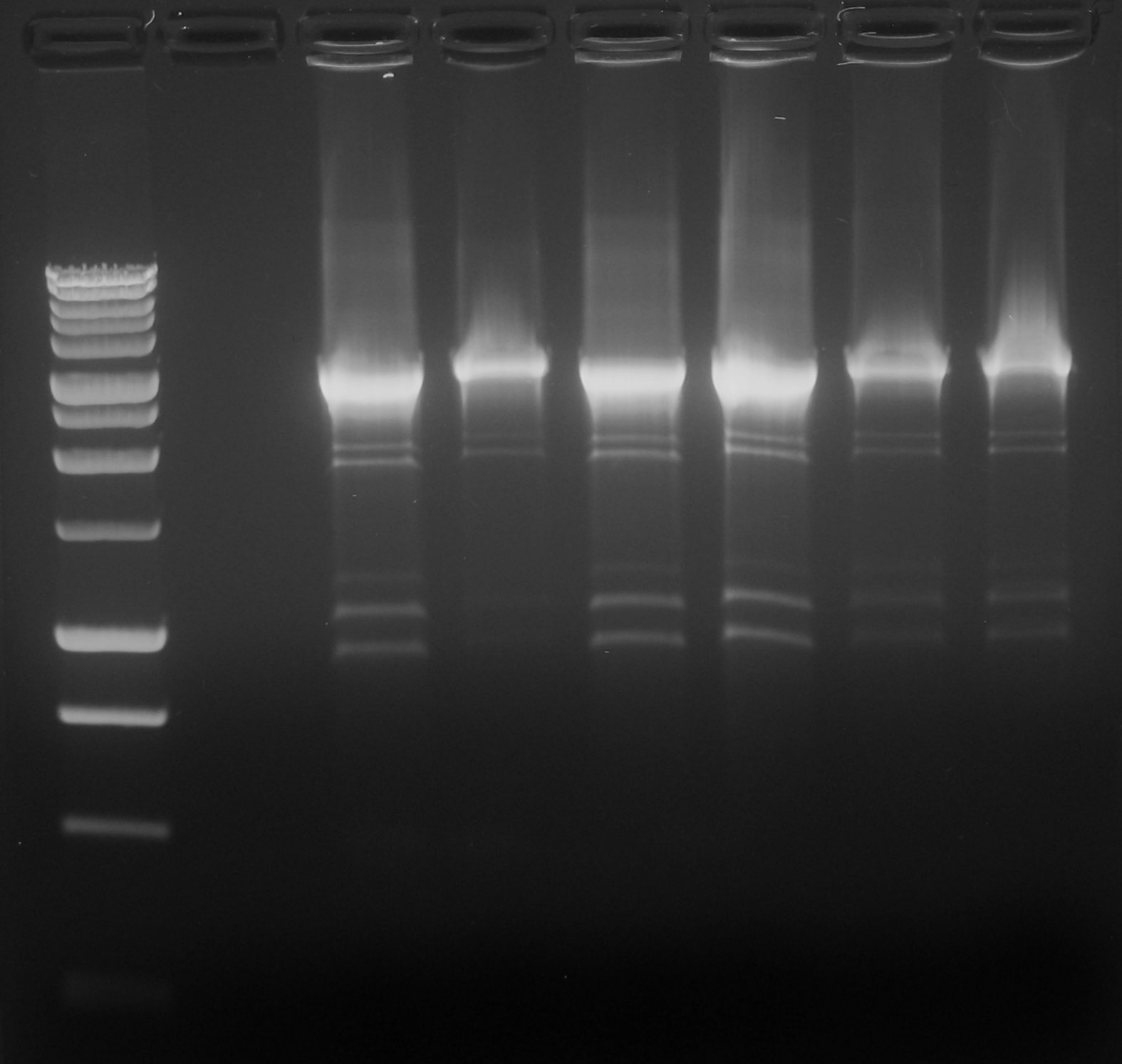
- We performed colony PCR on the two plates. Medium and large gel could not be ran because instrumentation was buisy. We could ran two small gels (8 wells), so the maximum number of colonies was 13 (considering two markers and one blank). Whe chose to screen 6 colonies for TT and 7 colonies for TR.
- Gel results:
- TT - 2nd colony
- TR - 1st colony (gel picture not available...sorry!)
- We incubated the chosen colonies at 37°C, 220 rpm overnight.
10/2/08
- Glycerol stocks for TT-2 and TR-1.
- TR fluorescence test. Experiment details and results are reported in The Project section, Experiments.
- Data processing.
10/3/08
- We prepared 0.5 l of LB + Amp for liquid cultures because our LB was looked turbid.
- Wiki updating.
 "
"