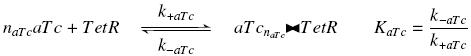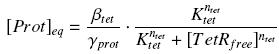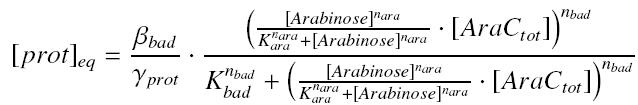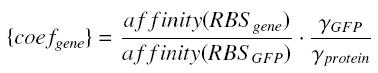Team:Paris/Modeling/estimation
From 2008.igem.org
(→what are we looking for ?) |
|||
| Line 1: | Line 1: | ||
{{Paris/Menu}} | {{Paris/Menu}} | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| + | =Estimation of parameters= | ||
If we want to use the promoters used for the formation of the flagella ([[Team:Paris/Project|Description of the project]]), we will have to clearly defined their dynamics. To do so, a rather huge experimental work will be undertaken, consisting in providing the so-called 'Hill functions' for each promoters. | If we want to use the promoters used for the formation of the flagella ([[Team:Paris/Project|Description of the project]]), we will have to clearly defined their dynamics. To do so, a rather huge experimental work will be undertaken, consisting in providing the so-called 'Hill functions' for each promoters. | ||
| - | == | + | ==Getting a Hill function from convenient datas== |
| - | + | ||
Therefore, we have written a little module which can estimate the parameters of the 'Hill functions', even with some noise and few data available. | Therefore, we have written a little module which can estimate the parameters of the 'Hill functions', even with some noise and few data available. | ||
Some details and the corresponding code can be found here : [[Team:Paris/Modeling/Programs|findparam]]. | Some details and the corresponding code can be found here : [[Team:Paris/Modeling/Programs|findparam]]. | ||
| Line 15: | Line 10: | ||
The method we have employed is just based on a least-square optimization. Then, it could be generic enough for many applications and we would be glad to share the code if you feel it could be usefull. | The method we have employed is just based on a least-square optimization. Then, it could be generic enough for many applications and we would be glad to share the code if you feel it could be usefull. | ||
| - | == | + | ==Getting convenient datas== |
| - | + | ||
Thus, we need experimental datas. To quantify the strength of a transcription factor on a promoter, we will use measurements of GFP fluorescence, and compare to the strength of the constitutive promoter [[http://partsregistry.org/Measurement/SPU/Learn J23101]], as it was proposed by the iGEM competition. | Thus, we need experimental datas. To quantify the strength of a transcription factor on a promoter, we will use measurements of GFP fluorescence, and compare to the strength of the constitutive promoter [[http://partsregistry.org/Measurement/SPU/Learn J23101]], as it was proposed by the iGEM competition. | ||
The datas we are looking for must appear as a table of values, giving several 'transduction rates' with their corresponding 'transcription factor concentrations'. | The datas we are looking for must appear as a table of values, giving several 'transduction rates' with their corresponding 'transcription factor concentrations'. | ||
| - | == | + | ==First hypothesis== |
For this aim, we made several hypothesis, which we will verify as good as it is possible for us : | For this aim, we made several hypothesis, which we will verify as good as it is possible for us : | ||
| Line 43: | Line 37: | ||
<br>Actually, we found a linear relation between GFP mut3B concentration (nM) and fluorescence (au). The conversion factor is 79,429. For more detailed information, see [[Team:Paris/Modeling/Bibliography|[6]]]. | <br>Actually, we found a linear relation between GFP mut3B concentration (nM) and fluorescence (au). The conversion factor is 79,429. For more detailed information, see [[Team:Paris/Modeling/Bibliography|[6]]]. | ||
| - | == | + | ==How to control the concentration of the transcription factor ?== |
| - | + | ===<center>By aTc / TetR / pTet </center>=== | |
| - | + | ||
| - | + | ||
Now, we must use as a variable of reference an element that could be introduced in the bacteria, well-controlled, and from which all the concentrations of our transcription factor will depend. We propose a construction in which our transcription factor is put after the promoter pTet, which is under the repression of TetR. Since aTc is a small diffusive molecule that binds to TetR and inhibits this way the repression of pTet, we can use it as an 'inducer'. To do so, we must place in the bacterium the gene ''tetR'' after a constitutive promoter (like J23101). According to previous hypothesis, this will provide at steady-state a 'constant concentration' of TetR (we note [TetR]<sub>tot</sub>, and it is supposed to be the TOTAL concentration of TetR, under every form) in the bacterium. If we consider the binding reaction this way (where aTc_-_TetR denotes the complex) | Now, we must use as a variable of reference an element that could be introduced in the bacteria, well-controlled, and from which all the concentrations of our transcription factor will depend. We propose a construction in which our transcription factor is put after the promoter pTet, which is under the repression of TetR. Since aTc is a small diffusive molecule that binds to TetR and inhibits this way the repression of pTet, we can use it as an 'inducer'. To do so, we must place in the bacterium the gene ''tetR'' after a constitutive promoter (like J23101). According to previous hypothesis, this will provide at steady-state a 'constant concentration' of TetR (we note [TetR]<sub>tot</sub>, and it is supposed to be the TOTAL concentration of TetR, under every form) in the bacterium. If we consider the binding reaction this way (where aTc_-_TetR denotes the complex) | ||
| Line 69: | Line 61: | ||
Then, we intend to program a new algorithm, based on the same principles of 'findparam' for a classic ''hill function'', but which is seeking more parameters. | Then, we intend to program a new algorithm, based on the same principles of 'findparam' for a classic ''hill function'', but which is seeking more parameters. | ||
| - | + | ===<center>By AraC / Arabinose / pBad </center>=== | |
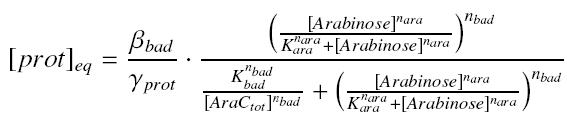
An other well-known promoter called pBad, is induced by the complex AraC_-_Arabinose, where AraC appears to be a protein ''constitutively produced'' by an operon attached to pBad, and Arabinose is a sugar, that we can add at will in the medium, and diffuses through cells. | An other well-known promoter called pBad, is induced by the complex AraC_-_Arabinose, where AraC appears to be a protein ''constitutively produced'' by an operon attached to pBad, and Arabinose is a sugar, that we can add at will in the medium, and diffuses through cells. | ||
| Line 87: | Line 79: | ||
and treat it exactly as for the pTet expression. | and treat it exactly as for the pTet expression. | ||
| - | + | ==The Problem of the RBS== | |
| - | + | ||
The promoter is not the only one factor which control the expression of the protein... in particular, the ''traduction phenomenon'' is almost as important as the ''transduction''. As we decided not to take into acount the traduction, that means that we do not want to deal with mRNA and ribosomes ; nevertheless, as we aimed in the "hill approach" to simulate as precisely as possible the involved concentrations, we must ''integrate in the traduction simulation the most important influences of the transduction'''. | The promoter is not the only one factor which control the expression of the protein... in particular, the ''traduction phenomenon'' is almost as important as the ''transduction''. As we decided not to take into acount the traduction, that means that we do not want to deal with mRNA and ribosomes ; nevertheless, as we aimed in the "hill approach" to simulate as precisely as possible the involved concentrations, we must ''integrate in the traduction simulation the most important influences of the transduction'''. | ||
| Line 113: | Line 104: | ||
To conclude, we nevertheless must notice that the effect of the coefficient γ in our model has got an other effect : it changes the dynamics of the system, in the time in which the ''steady states'' are reached. As our system intend to produce ''oscillations'', the ''transcient states'' are important, and here is the biggest weakness of this model. | To conclude, we nevertheless must notice that the effect of the coefficient γ in our model has got an other effect : it changes the dynamics of the system, in the time in which the ''steady states'' are reached. As our system intend to produce ''oscillations'', the ''transcient states'' are important, and here is the biggest weakness of this model. | ||
| - | == | + | ==What are we looking for ?== |
| - | + | ||
The different functions we would like to determine are the followings. They are linked to a basic description of the "theoretical protocol" that will allow us to get the expected datas. We decide to let the original promoters in the bacteria, so that the strength that we are measuring is "the strength for an '''additional''' promoter in the cell", keeping those which already exist : this makes sense for our construction, and probably for most of the constructions of synthetic biology wih ''E.coli''. | The different functions we would like to determine are the followings. They are linked to a basic description of the "theoretical protocol" that will allow us to get the expected datas. We decide to let the original promoters in the bacteria, so that the strength that we are measuring is "the strength for an '''additional''' promoter in the cell", keeping those which already exist : this makes sense for our construction, and probably for most of the constructions of synthetic biology wih ''E.coli''. | ||
| Line 145: | Line 135: | ||
*[[Team:Paris/Modeling/f9|[expr.(pLas)] = ƒ9([LasR_-_HSL])]] | *[[Team:Paris/Modeling/f9|[expr.(pLas)] = ƒ9([LasR_-_HSL])]] | ||
| - | + | = Noise estimation = | |
| + | Firstly, we could compute the standard deviation for each set of points at a given inducer concentration, and to normalize it according to the mean value of the set itself. | ||
| + | |||
| + | The next stage is to get the mean of those normalized sd values at every inducer concentration. | ||
| + | |||
| + | Since the β parameter of the Hill function has a linear influence, it is possible to translate this error directly on the parameter; for example: '''β_err = random('norm',β,err)''' where '''β''' is the estimated value of the parameter and '''err''' is the mean of the normalized standard deviations of the experimental values. | ||
| + | |||
| + | For each cell in the model, we could use such noised values for Vmax parameter, in order to reproduce randomness estimated in the wet lab. | ||
Revision as of 15:35, 27 August 2008
Estimation of parametersIf we want to use the promoters used for the formation of the flagella (Description of the project), we will have to clearly defined their dynamics. To do so, a rather huge experimental work will be undertaken, consisting in providing the so-called 'Hill functions' for each promoters. Getting a Hill function from convenient datasTherefore, we have written a little module which can estimate the parameters of the 'Hill functions', even with some noise and few data available. Some details and the corresponding code can be found here : findparam. The method we have employed is just based on a least-square optimization. Then, it could be generic enough for many applications and we would be glad to share the code if you feel it could be usefull. Getting convenient datasThus, we need experimental datas. To quantify the strength of a transcription factor on a promoter, we will use measurements of GFP fluorescence, and compare to the strength of the constitutive promoter http://partsregistry.org/Measurement/SPU/Learn J23101, as it was proposed by the iGEM competition. The datas we are looking for must appear as a table of values, giving several 'transduction rates' with their corresponding 'transcription factor concentrations'. First hypothesisFor this aim, we made several hypothesis, which we will verify as good as it is possible for us : (1) We do not take into acount the 'traduction' phase (see however considerations on RBS), so we directly correlate the transcription of a gene with the concentration of its protein. (2) We assume that, whatever is the gene behind the promoter, its expression depends only of the transcription factor of the promoter, and not, for instance, of the weight of this gene. That's why comparing promoter strength is relevent only if the genes behind have similar length. (3) We consider that the activity of a promoter is well described as a Hill function of its transcription factor (TF). Thus, we suppose that the protein concentration (Prot) follows this equation : 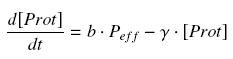 where gamma is a constant, due to degradation and of dilution of the protein, along time and cell divisions. Therefore, if we consider a steady-state, for given concentration of the transcription factor, we will have : (4) Endly, knowing γ will give us the kind of datas we are looking for. In a first approach, we assume that, as long as the barcteria are in their phase of exponential growth, the degradation is far smaller than the dilution, and can be omitted. But we will probably discuss that later (see considerations on RBS). We assume that in the phase of exponential growth (that we manage to keep permanent with the system of Ron Weiss, see our project), each cells divide every hour ; that makes a half-life time of 3600 s, and a coefficient of dilution equal to ln(2)/3600 s-1. (5) Unless we find further documents dealing with the relation between the intensity of fluorescence and the concentration of GFP, we will directly use the measure in fluorescence, that we will treat as a protein concentration, more or less arbitrary normalised.
How to control the concentration of the transcription factor ?
|
 "
"