Team:Paris/August 6
From 2008.igem.org
(→Cloning of pflhDC) |
|||
| Line 143: | Line 143: | ||
===Cloning of pflhDC=== | ===Cloning of pflhDC=== | ||
| - | [Team:Paris/August_5#Electrophoresis Purification of PCR Yesterday] | + | [[Team:Paris/August_5#Electrophoresis Purification of PCR|Yesterday]] the isolation of pflhDC did not work : the PCR result measured more than 1.5 kb. We checked the primers, they are well designed but there is a sequence homology with other sequences in ''E.coli'' K12. |
| + | To amplify more precisely the promoter, we decided to do a PCR with gradient. | ||
| + | |||
| + | ====Protocol==== | ||
| + | |||
| + | *10µL Phusion buffer 5X | ||
| + | *1µL dNTP | ||
| + | *2.5 µL Oligo F (O111) 10mM | ||
| + | *2.5 µL Oligo R (O113) 10mM | ||
| + | *1µL template DNA | ||
| + | *0.5µL Phusion polymerase | ||
| + | |||
| + | ====PCR Program==== | ||
| + | |||
| + | '''PROMOTE2''' | ||
| + | * LID : 105 °C | ||
| + | *1. T: 95°C 5min | ||
| + | *2. T: 95°C 5min | ||
| + | *3. T: 55°C 30s ~>G: 5°C | ||
| + | *4. T: 72°C 1min30 | ||
| + | *6. GO TO: 2 REP: 29 | ||
| + | *7. HOLD: 10°C | ||
| + | |||
| + | ====Results==== | ||
| + | |||
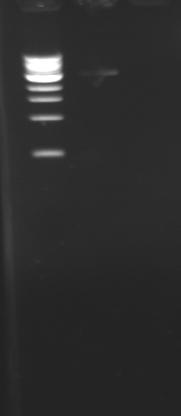
| + | [[Image:KR000113.jpg|thumb|PCR with gradient : amplification of pflhDC]] | ||
Revision as of 18:47, 6 August 2008
Assay to clone different flagella gene promotors into the J61002 plasmidWe have to digest (by EcoRI and SpeI) the PCR products of yesterday (amplification of pFlgA (MP124), pFlgB (MP125), pFlhB (MP126) and pFlhDC (MP127) in order to clone them into the J61002 plasmid (that we must extract and then digest by EcoRI and SpeI).
Plasmid extractionThe J61002 plasmid was extracted from overnight bacteria culture using the QIAspin Miniprep Kit (QIAGEN).
Assay to quantify DNAIn order to quantify the DNA contained either in the Miniprep product of MP123 or in the PCR products of MP124, MP125, MP126 and MP127 previously purifed yesterday by the QIAquick Gel Extraction Kit, we carried out an electrophoresis assay in a 1,5% agarose gel.
Results : The MP123 plasmid is clearly visible but the PCR products (purified by QIAquick Gel Extraction) aren't. There might be a problem during the purification step using the QIAquick Gel Extraction. Whatever, we still go on with the digestion.
DigestionFor each sample (MP123 and PCR products of MP124, MP125, MP126 and MP127):
=> Concentration : +/- 12ng/4µL -> 3ng/µL LigationProtocolFor each samples,
List of ligations
Cloning of pflhDCYesterday the isolation of pflhDC did not work : the PCR result measured more than 1.5 kb. We checked the primers, they are well designed but there is a sequence homology with other sequences in E.coli K12. To amplify more precisely the promoter, we decided to do a PCR with gradient. Protocol
PCR ProgramPROMOTE2
Results |
 "
"