Team:Paris/August 26
From 2008.igem.org
(→Cloning of EnvZ*) |
(→PCR) |
||
| Line 102: | Line 102: | ||
| - | =PCR= | + | =PCR Promoters and Genes FlhDC/FliA= |
| - | == | + | ==PCR Promoters FlhDC and Gene== |
* pFlhDC (O111-F / O113-R) | * pFlhDC (O111-F / O113-R) | ||
| Line 135: | Line 135: | ||
Phusion=0,2µL<br> | Phusion=0,2µL<br> | ||
| - | == | + | ==PCR Promoters FliA== |
* pFliA (rbs) (O145-F / O144-R) | * pFliA (rbs) (O145-F / O144-R) | ||
| Line 166: | Line 166: | ||
Phusion=0,5µL<br> | Phusion=0,5µL<br> | ||
| - | == | + | ==PCR mutagenesis FliA== |
| - | * PCRFliA1 (O143-F / O152-R) | + | * PCRFliA1 (O143-F / O152-R) - PCRFliA1' (O148-F / O152-R) |
* PCRFliA2 (O153-F / O150-R) | * PCRFliA2 (O153-F / O150-R) | ||
* PCRFliA3 (O148-F / O150-R) | * PCRFliA3 (O148-F / O150-R) | ||
| Line 180: | Line 180: | ||
98°C-10"<br> | 98°C-10"<br> | ||
Gradient 66°C +/-6°C - 25''<br> | Gradient 66°C +/-6°C - 25''<br> | ||
| + | 72°C-20"<br> | ||
| + | '''''Elongation :''''' <br> | ||
| + | 72°C-5'<br> | ||
| + | |||
| + | '''Program: PCRFliA1''''<br> | ||
| + | '''''Denaturation :''''' <br> | ||
| + | 98°C-5'<br> | ||
| + | '''''Cycling 1 (30X) :''''' <br> | ||
| + | 98°C-10"<br> | ||
72°C-20"<br> | 72°C-20"<br> | ||
'''''Elongation :''''' <br> | '''''Elongation :''''' <br> | ||
| Line 200: | Line 209: | ||
'''''Denaturation :''''' <br> | '''''Denaturation :''''' <br> | ||
98°C-30'<br> | 98°C-30'<br> | ||
| - | '''''Cycling 1 ( | + | '''''Cycling 1 (3X) :''''' <br> |
98°C-10"<br> | 98°C-10"<br> | ||
72°C-30"<br> | 72°C-30"<br> | ||
| Line 206: | Line 215: | ||
98°C-10"<br> | 98°C-10"<br> | ||
98°C->72°C low decreasing 1.0°C/s<br> | 98°C->72°C low decreasing 1.0°C/s<br> | ||
| + | 72°C-30"<br> | ||
| + | '''''Break - Add Oligo O148/O150'''''<br> | ||
| + | '''''Cycling 3 (20X) :'''''<br> | ||
| + | 98°C-10"<br> | ||
72°C-30"<br> | 72°C-30"<br> | ||
'''''Elongation :''''' <br> | '''''Elongation :''''' <br> | ||
| Line 222: | Line 235: | ||
|2-27 | |2-27 | ||
|197 bp | |197 bp | ||
| - | |style="background: #cbff7B"| ~ | + | |style="background: #cbff7B"| ~ 150 bp |
| + | |- | ||
| + | |PCRFliA2 | ||
| + | |Downstream part of FliA | ||
| + | |28-54 | ||
| + | |670 bp | ||
| + | |style="background: #cbff7B"| ~ 650 bp | ||
|- | |- | ||
|PCRFliA2 | |PCRFliA2 | ||
|Downstream part of FliA | |Downstream part of FliA | ||
|28-54 | |28-54 | ||
| - | | | + | |800 bp |
| - | |style="background: #cbff7B"| | + | |style="background: #cbff7B"| ~ 800 bp |
|- | |- | ||
|PCR | |PCR | ||
Revision as of 14:35, 27 August 2008
Extraction of EnvZ* and OmpR* from E. coli genomeName of the PCR
ProtocolProtocol
PCR Programme
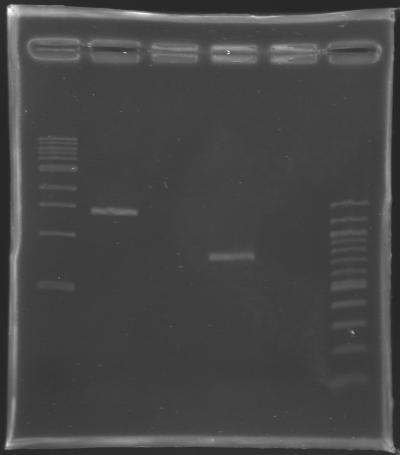
ResutsElectrophoresis settings
Results of the electrophoresis
Conclusion : All the PCR worked perfectly well ! Cleaning of the PCR productsThe cleaned PCR products are stored in the freezer overnight.
PCR Promoters and Genes FlhDC/FliAPCR Promoters FlhDC and Gene
Program: Gradient Vf=20µL PCR Promoters FliA
Program: promoter Vf=50µL PCR mutagenesis FliA
Program: PCRFliA1 Program: PCRFliA1' Program: PCRFliA2 Program: PCRFliA3
Cloning of EnvZ*Concentration measurement by Biophotometer
Ligation
Miniprep and stock glycerolof New Biobrick
Construction of pFlhB - mRFP Tripart (LVA+)Aim : Construction of "pFlhB-RBS-mRFP-dbl ter" (pFlhB-I732078) DigestionDigestion
Gel VerificationFile:KR000.JPG Gel Verification of D187 digestion
Screening of the cloning of pFlgA-GFP GeneratorElectrophoresis
File:KR000.jpg Screening of L164 Minipreps and glycerol stock
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"