Team:Paris/July 29
From 2008.igem.org
(→DNA digestion and purification) |
(→DNA digestion and purification) |
||
| Line 35: | Line 35: | ||
! rowspan="1" style="background: #ccccff;" | D101 | ! rowspan="1" style="background: #ccccff;" | D101 | ||
! rowspan="1" style="background: #ccccff;" | B0034 | ! rowspan="1" style="background: #ccccff;" | B0034 | ||
| - | ! rowspan="1"| | + | ! rowspan="1"|1 |
! rowspan="1"| EcoRI | ! rowspan="1"| EcoRI | ||
! rowspan="1"| XbaI | ! rowspan="1"| XbaI | ||
| Line 47: | Line 47: | ||
! rowspan="1" style="background: #ccccff;" | D102 | ! rowspan="1" style="background: #ccccff;" | D102 | ||
! rowspan="1" style="background: #ccccff;" | B0034 | ! rowspan="1" style="background: #ccccff;" | B0034 | ||
| - | ! rowspan="1"| | + | ! rowspan="1"|1 |
! rowspan="1"| SpeI | ! rowspan="1"| SpeI | ||
! rowspan="1"| PstI | ! rowspan="1"| PstI | ||
| Line 59: | Line 59: | ||
! rowspan="1" style="background: #ccccff;" | D105 | ! rowspan="1" style="background: #ccccff;" | D105 | ||
! rowspan="1" style="background: #ccccff;" | R0079 | ! rowspan="1" style="background: #ccccff;" | R0079 | ||
| - | ! rowspan="1"| | + | ! rowspan="1"|1 |
! rowspan="1"| SpeI | ! rowspan="1"| SpeI | ||
! rowspan="1"| PstI | ! rowspan="1"| PstI | ||
| Line 71: | Line 71: | ||
! rowspan="1" style="background: #ccccff;" | D106 | ! rowspan="1" style="background: #ccccff;" | D106 | ||
! rowspan="1" style="background: #ccccff;" | R0040 | ! rowspan="1" style="background: #ccccff;" | R0040 | ||
| - | ! rowspan="1"| | + | ! rowspan="1"|1 |
! rowspan="1"| SpeI | ! rowspan="1"| SpeI | ||
! rowspan="1"| PstI | ! rowspan="1"| PstI | ||
| Line 83: | Line 83: | ||
! rowspan="1" style="background: #ccccff;" | D108 | ! rowspan="1" style="background: #ccccff;" | D108 | ||
! rowspan="1" style="background: #ccccff;" | S03154 | ! rowspan="1" style="background: #ccccff;" | S03154 | ||
| - | ! rowspan="1"| | + | ! rowspan="1"|1 |
! rowspan="1"| XbaI | ! rowspan="1"| XbaI | ||
! rowspan="1"| PstI | ! rowspan="1"| PstI | ||
| Line 95: | Line 95: | ||
! rowspan="1" style="background: #ccccff;" | D111 | ! rowspan="1" style="background: #ccccff;" | D111 | ||
! rowspan="1" style="background: #ccccff;" | S03879 | ! rowspan="1" style="background: #ccccff;" | S03879 | ||
| - | ! rowspan="1"| | + | ! rowspan="1"|1 |
! rowspan="1"| XbaI | ! rowspan="1"| XbaI | ||
! rowspan="1"| PstI | ! rowspan="1"| PstI | ||
| Line 131: | Line 131: | ||
! rowspan="1" style="background: #ccccff;" | D117 | ! rowspan="1" style="background: #ccccff;" | D117 | ||
! rowspan="1" style="background: #ccccff;" | E0030 | ! rowspan="1" style="background: #ccccff;" | E0030 | ||
| - | ! rowspan="1"| | + | ! rowspan="1"|1 |
! rowspan="1"| EcoRI | ! rowspan="1"| EcoRI | ||
! rowspan="1"| SpeI | ! rowspan="1"| SpeI | ||
Revision as of 14:16, 31 July 2008
DNA digestion and purificationfor each reaction (total volume : 50 µL)
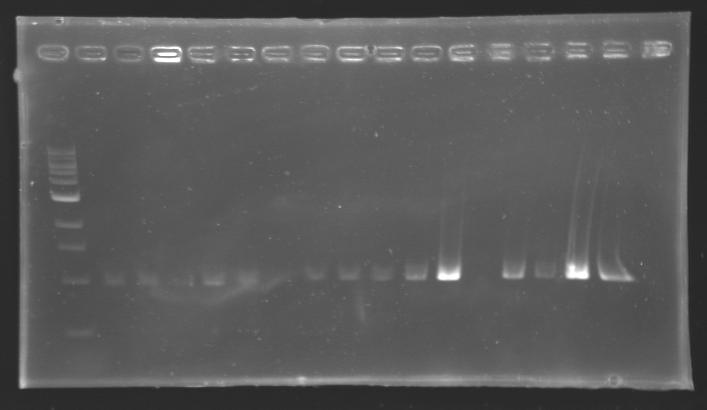
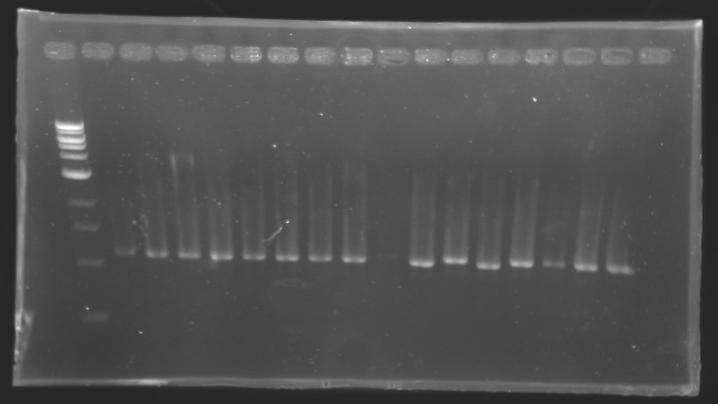
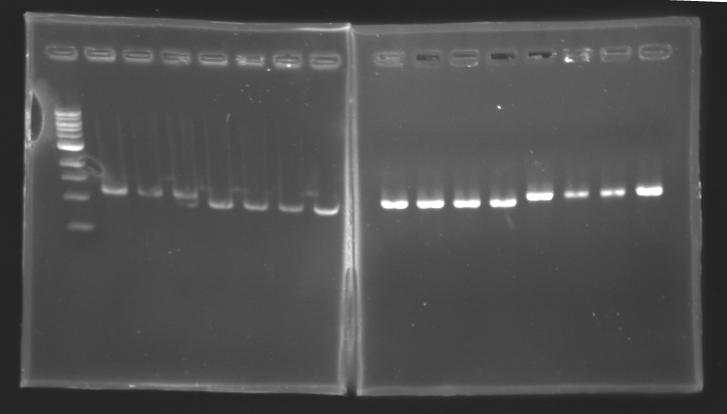
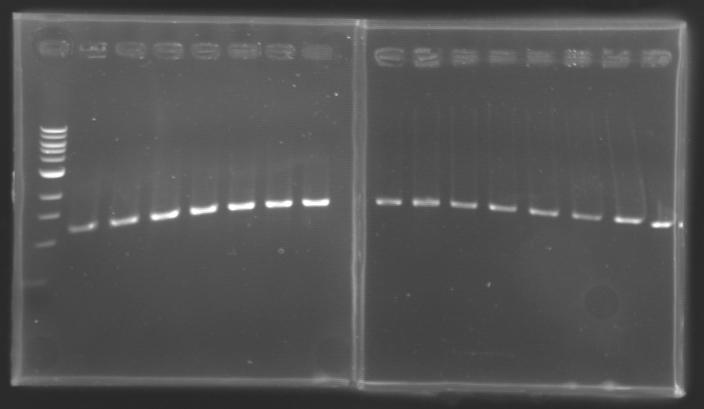
Each reaction was incubated 2 hours at 37°C, then 10 minutes at 60-65°C (to inactivate the enzymes). 10 µL of loading dye (6X) were added to each of the 50 µL of digestion product. The whole samples were run in a 1,5% agarose gel (about 30 minutes at 100 W ; 2 x 30 µL per sample ; 30 µL per well). The bands of interest were then excised from the gel and the DNA was purified using the QIAquick DNA Gel Extraction kit (QIAGEN). Unfortunately, there were not enough columms, so we took some columms from the QIAGEN MiniPrep kit, hoping that it will work with the QIAquick DNA Gel Extraction kit. Some of the samples were too voluminous, so we separated them into two tubes. The elution of DNA was performed using 50 µL of water (after 10 minutes of incubation at 37°C). Each of the samples was then analysed by a 1,5% agarose gel:
The ladder used was the 100 bp ladder from New England Biolabs.
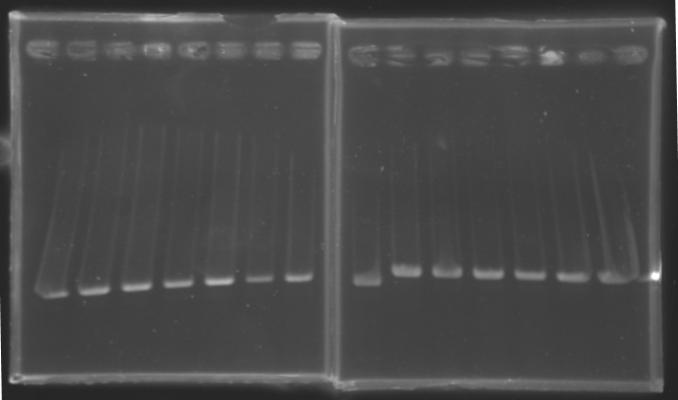
Results : The amount of DNA loaded was too low and the DNA ladder used was the wrong one. So this experiment has to be reconducted tomorrow. PCR Screening of the Ligation TransformantsUse of 8 clones of Ligation transformants for screening PCR
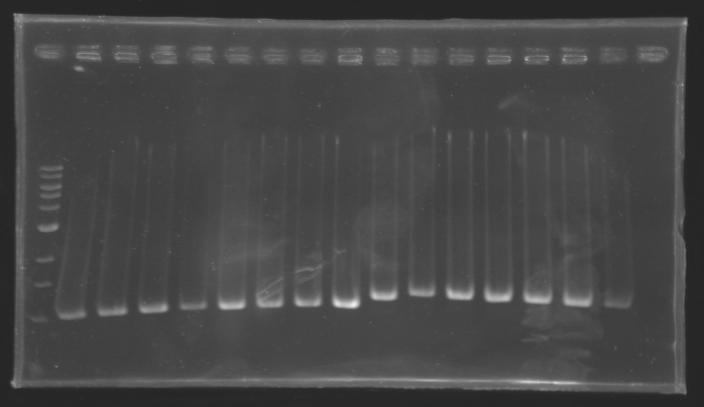
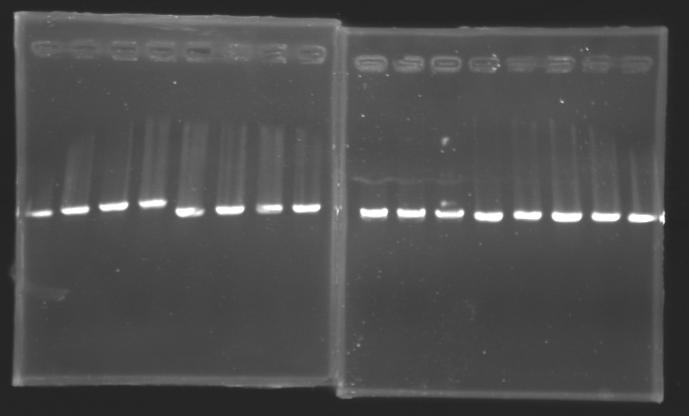
Protocol of screening PCR
Conditions of migration
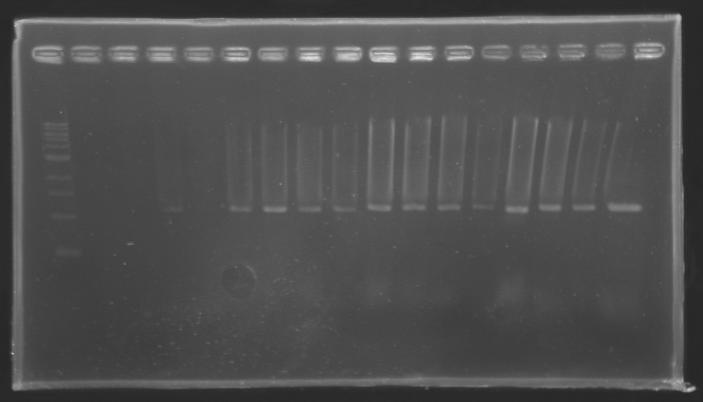
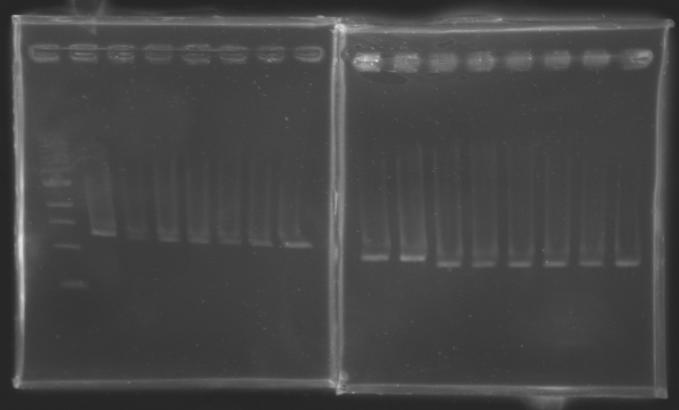
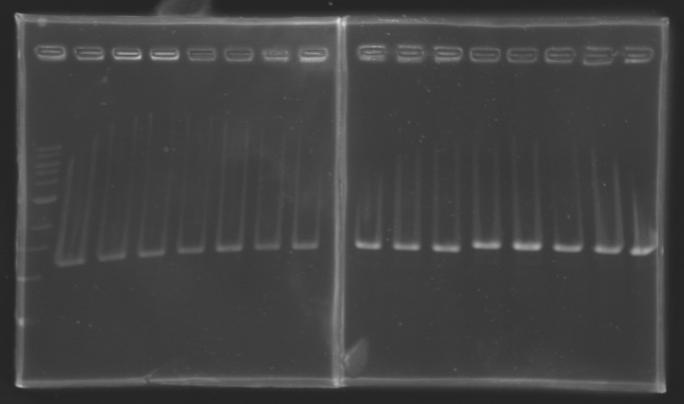
Results
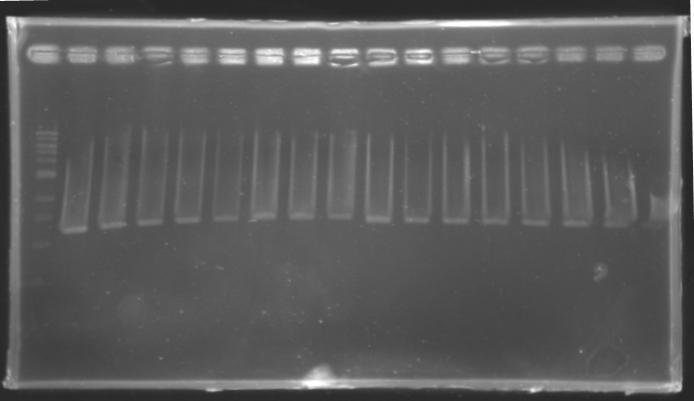
But we don't observe results for L102(3), L102(6), L103(4), L106(1), L106(2), L106(4), L111(1) Migration of an another gel for this sample...
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"