Team:Paris/July 24
From 2008.igem.org
(Difference between revisions)
(→Results of digestions : Electrophoresis) |
(→MiniPreps) |
||
| (20 intermediate revisions not shown) | |||
| Line 4: | Line 4: | ||
== MiniPreps == | == MiniPreps == | ||
| - | *The | + | *The Promega MiniPreps protocol has been used on all the clones cultivated on the 23th. |
{| border="1" | {| border="1" | ||
| Line 11: | Line 11: | ||
|align="center"|'''Description''' | |align="center"|'''Description''' | ||
|- | |- | ||
| - | |align="center"|MP116 | + | |align="center"|MP116 |
| - | |align="center"|J23100 | + | |align="center"|[http://partsregistry.org/Part:BBa_J23100 J23100] |
| - | |align="center"| | + | |align="center"|Strong constitutive promoter in J61002 |
|- | |- | ||
| - | |align="center"|MP117 | + | |align="center"|MP117 |
| - | |align="center"|J23107 | + | |align="center"|[http://partsregistry.org/Part:BBa_J23107 J23107] |
| - | |align="center"| | + | |align="center"|Medium constitutive promoter in J61002 |
|- | |- | ||
| - | |align="center"|MP118 | + | |align="center"|MP118 |
| - | |align="center"|B0015 | + | |align="center"|[http://partsregistry.org/Part:BBa_B0015 B0015] |
| - | |align="center"| | + | |align="center"|Double terminator |
|- | |- | ||
| - | |align="center"|MP119 | + | |align="center"|MP119 |
| - | |align="center"|I0500 | + | |align="center"|[http://partsregistry.org/Part:BBa_I0500 I0500] |
| - | |align="center"| | + | |align="center"|AraC pBAD |
|- | |- | ||
| - | |align="center"|MP120 | + | |align="center"|MP120 |
| - | |align="center"|B0030 | + | |align="center"|[http://partsregistry.org/Part:BBa_B0030 B0030] |
| - | |align="center"| | + | |align="center"|Strong RBS (Efficiency = 0,6) |
|- | |- | ||
| - | |align="center"|MP121 | + | |align="center"|MP121 |
| - | |align="center"|E0422 | + | |align="center"|[http://partsregistry.org/Part:BBa_E0422 E0422] |
| - | |align="center"| | + | |align="center"|ECFP (RBS+LVA+Term) |
|- | |- | ||
| - | |align="center"|MP122 | + | |align="center"| MP122 |
| - | |align="center"|E0840 | + | |align="center"|[http://partsregistry.org/Part:BBa_E0840 E0840] |
| - | |align="center"| | + | |align="center"|gfp tri-part; strong rbs |
| + | | | ||
|} | |} | ||
| - | |||
| - | |||
| - | |||
== Digestion == | == Digestion == | ||
| - | |||
===Digestion Mix=== | ===Digestion Mix=== | ||
| - | 10µl of Miniprep ( | + | 10µl of Miniprep (26 aug.) <br> |
12.5µl of water<br> | 12.5µl of water<br> | ||
2.5µl of Buffer N°2<br> | 2.5µl of Buffer N°2<br> | ||
| Line 56: | Line 53: | ||
1µl of enzyme 2<br> | 1µl of enzyme 2<br> | ||
| - | + | * Incubation 1h at 37°C with the first enzyme | |
| + | * Add the second enzyme | ||
| + | * Incubation 1h at 37°C with the second enzyme | ||
| + | * Store on ice | ||
| + | * Revelation of the digestion by electrophoresis on agarose gel | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
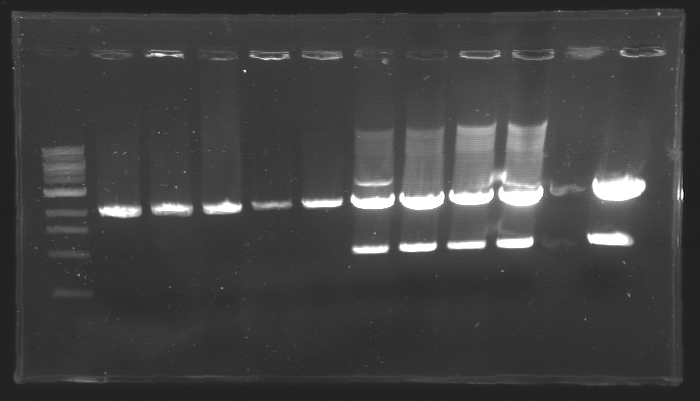
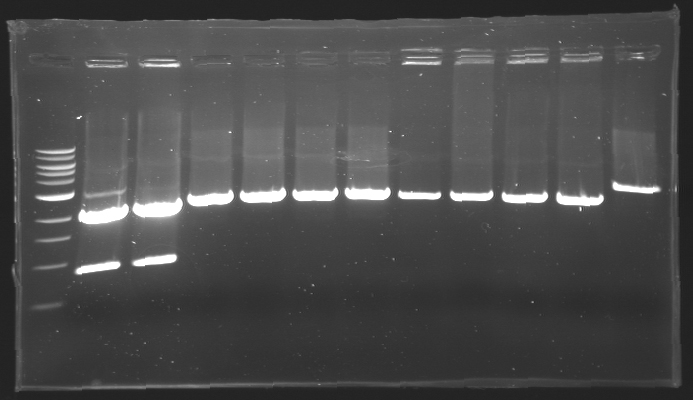
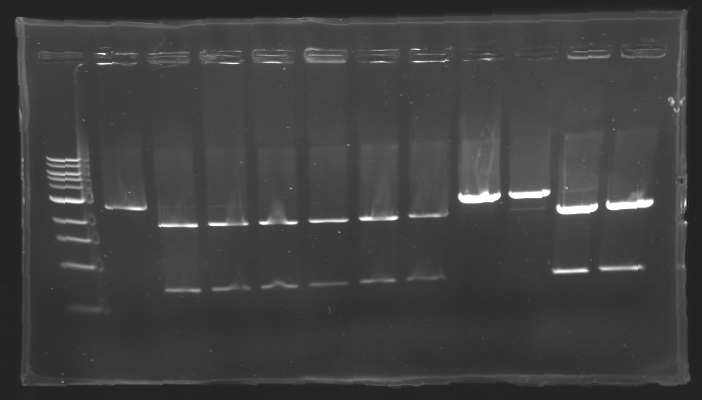
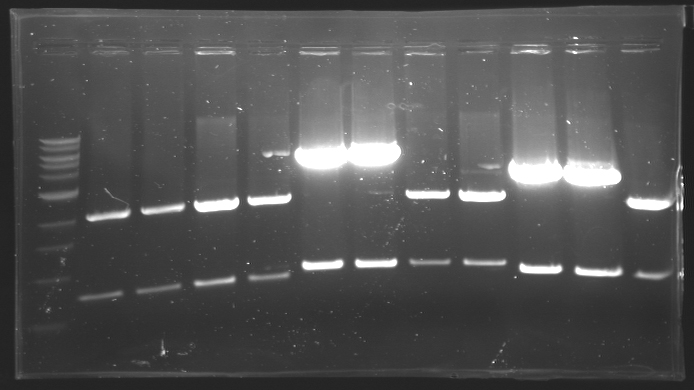
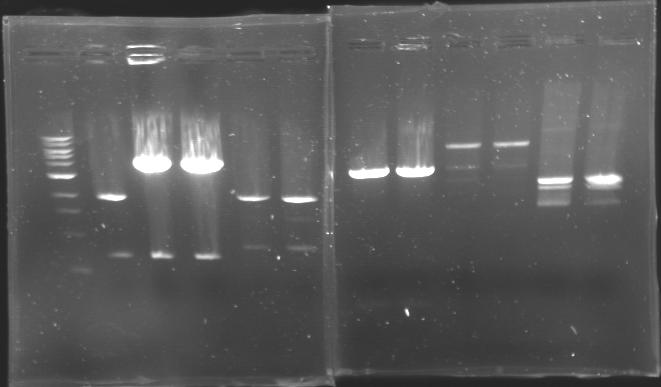
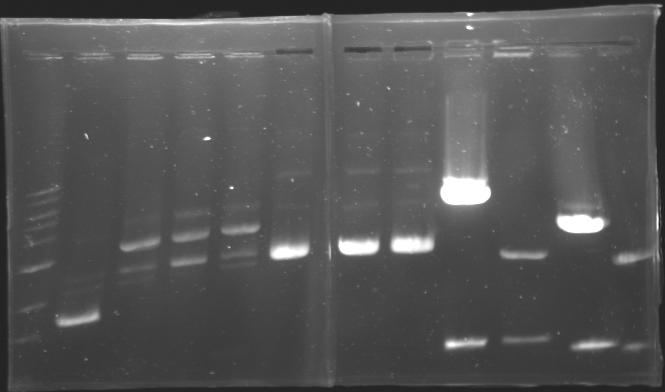
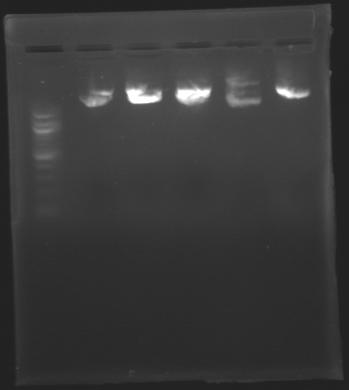
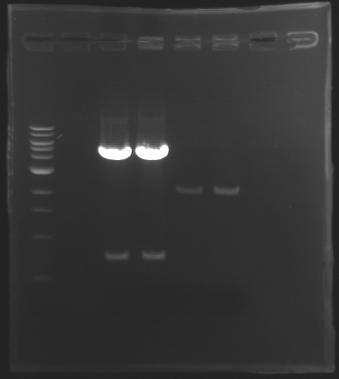
| - | == Results of digestions : Electrophoresis == | + | === Results of digestions : Electrophoresis === |
| + | |||
| + | conditions : | ||
| + | * 10µl of ladder 1 kb (except for gel n°7 : 100 pb) | ||
| + | * 30µl of digestion added with 5µl of loading Dye 6x | ||
| + | * migration ~30min at 100W | ||
| + | * Gel 1, 2, 3, 4, 5, 6, 8 = '''0.8%''' | ||
| + | * Gel 7 = '''1,2%''' | ||
| + | |||
| + | |||
| + | |||
| + | gel1 [[Image:Gel1.jpg| gel1|200px]] | ||
| + | gel2 [[Image:Gel2.jpg| gel2|200px]] | ||
| + | gel3 [[Image:Gel3.jpg| gel3|200px]] | ||
| + | gel4 [[Image:Gel4.jpg| gel4|200px]]<br> | ||
| + | |||
| + | |||
| + | gel5 [[Image:Gel_5.jpg| gel5|200px]] | ||
| + | gel6 [[Image:Gel_6.jpg| gel6|200px]] | ||
| + | gel7 [[Image:Gel_7.jpg| gel7|100px]] | ||
| + | gel8 [[Image:Gel_8.jpg| gel8|100px]] | ||
| + | |||
| + | |||
{| border="1" | {| border="1" | ||
| Line 283: | Line 109: | ||
! rowspan="2"| not digested | ! rowspan="2"| not digested | ||
! rowspan="2"| not digested | ! rowspan="2"| not digested | ||
| - | + | ! rowspan="2"| 8 | |
| - | + | ! rowspan="2"| 5-6 | |
|- | |- | ||
|align=center|2 | |align=center|2 | ||
| - | |||
| - | |||
|- | |- | ||
|align=center|3 | |align=center|3 | ||
| Line 330: | Line 154: | ||
|- | |- | ||
|align=center|2 | |align=center|2 | ||
| - | |||
| - | |||
|align=center| 1 | |align=center| 1 | ||
|align=center| 9-10 | |align=center| 9-10 | ||
| Line 398: | Line 220: | ||
! rowspan="2"| XbaI | ! rowspan="2"| XbaI | ||
! rowspan="2"| PstI | ! rowspan="2"| PstI | ||
| - | ! rowspan="2"| | + | ! rowspan="2"| BI |
! rowspan="2"| 2057 pb | ! rowspan="2"| 2057 pb | ||
! rowspan="2"| 707 pb | ! rowspan="2"| 707 pb | ||
| Line 413: | Line 235: | ||
| align=center|EcoRI | | align=center|EcoRI | ||
| align=center|SpeI | | align=center|SpeI | ||
| - | | align=center| | + | | align=center|FI |
| align=center| 2056 pb | | align=center| 2056 pb | ||
| align=center| 708 pb | | align=center| 708 pb | ||
| Line 437: | Line 259: | ||
! rowspan="2"| XbaI | ! rowspan="2"| XbaI | ||
! rowspan="2"| PstI | ! rowspan="2"| PstI | ||
| - | ! rowspan="2"| | + | ! rowspan="2"| BI |
! rowspan="2"| 2057 pb | ! rowspan="2"| 2057 pb | ||
! rowspan="2"| 725 pb | ! rowspan="2"| 725 pb | ||
| Line 452: | Line 274: | ||
| align=center|EcoRI | | align=center|EcoRI | ||
| align=center|SpeI | | align=center|SpeI | ||
| - | | align=center| | + | | align=center|FI |
| align=center| 2756 pb | | align=center| 2756 pb | ||
| align=center| 726 pb | | align=center| 726 pb | ||
| Line 481: | Line 303: | ||
|align=center| 5000 pb | |align=center| 5000 pb | ||
|align=center| 100 pb | |align=center| 100 pb | ||
| + | |align=center| 6 | ||
| + | |align=center| 9 | ||
|- | |- | ||
! rowspan="2" style="background: #ccccff;"|D108 | ! rowspan="2" style="background: #ccccff;"|D108 | ||
| Line 503: | Line 327: | ||
|align=center| 2500 pb | |align=center| 2500 pb | ||
|align=center| 1000 pb | |align=center| 1000 pb | ||
| + | |align=center| 6 | ||
| + | |align=center| 10 | ||
|- | |- | ||
! rowspan="2" style="background: #ccccff;"|D109 | ! rowspan="2" style="background: #ccccff;"|D109 | ||
| Line 525: | Line 351: | ||
|align=center| 3000 pb | |align=center| 3000 pb | ||
|align=center| 1000 pb | |align=center| 1000 pb | ||
| + | |align=center| 6 | ||
| + | |align=center| 11 | ||
|- | |- | ||
! rowspan="2" style="background: #ccccff;"|D110 | ! rowspan="2" style="background: #ccccff;"|D110 | ||
| Line 547: | Line 375: | ||
|align=center| 2500 pb | |align=center| 2500 pb | ||
|align=center| 1000 pb | |align=center| 1000 pb | ||
| + | |align=center| 6 | ||
| + | |align=center| 12 | ||
|- | |- | ||
! rowspan="2" style="background: #ccccff;"|D111 | ! rowspan="2" style="background: #ccccff;"|D111 | ||
| Line 569: | Line 399: | ||
|align=center| 4500 pb | |align=center| 4500 pb | ||
|align=center| 700 pb | |align=center| 700 pb | ||
| + | |align=center| 8 | ||
| + | |align=center| 2-3 | ||
|- | |- | ||
! rowspan="1" style="background: #ccccff;"|D116 | ! rowspan="1" style="background: #ccccff;"|D116 | ||
| Line 578: | Line 410: | ||
! rowspan="1"| 2100 pb | ! rowspan="1"| 2100 pb | ||
! rowspan="1"| 883 pb | ! rowspan="1"| 883 pb | ||
| - | ! colspan=" | + | ! colspan="4"| Not realised |
|align=center| | |align=center| | ||
|- | |- | ||
| Line 591: | Line 423: | ||
|align=center| 2000 pb | |align=center| 2000 pb | ||
|align=center| 800 pb | |align=center| 800 pb | ||
| + | |align=center| 5 | ||
| + | |align=center| 5-6 | ||
|- | |- | ||
! rowspan="1" style="background: #ccccff;"|D118 | ! rowspan="1" style="background: #ccccff;"|D118 | ||
| Line 602: | Line 436: | ||
|align=center| 3000 pb | |align=center| 3000 pb | ||
|align=center| - | |align=center| - | ||
| + | |align=center| 5 | ||
| + | |align=center| 7-8 | ||
|- | |- | ||
! rowspan="1" style="background: #ccccff;"|D119 | ! rowspan="1" style="background: #ccccff;"|D119 | ||
| Line 613: | Line 449: | ||
|align=center| 6000 - 3000 pb | |align=center| 6000 - 3000 pb | ||
|align=center| - | |align=center| - | ||
| + | |align=center| 5 | ||
| + | |align=center| 9-10 | ||
|- | |- | ||
! rowspan="5" style="background: #ccccff;" | D120 | ! rowspan="5" style="background: #ccccff;" | D120 | ||
| Line 624: | Line 462: | ||
! rowspan="2"| not digested | ! rowspan="2"| not digested | ||
! rowspan="2"| not digested | ! rowspan="2"| not digested | ||
| + | |align=center| 7 | ||
| + | |align=center| 2 | ||
|- | |- | ||
|align=center|2 | |align=center|2 | ||
| - | |align=center| | + | |align=center| 7 |
| - | |align=center| | + | |align=center| 5-6 |
|- | |- | ||
|align=center|3 | |align=center|3 | ||
| Line 637: | Line 477: | ||
|align=center| 1600 pb | |align=center| 1600 pb | ||
|align=center| - | |align=center| - | ||
| + | |align=center| 6 | ||
| + | |align=center| 2 | ||
|- | |- | ||
|align=center|4 | |align=center|4 | ||
| Line 646: | Line 488: | ||
! rowspan="2"| 3000 - 2000 pb | ! rowspan="2"| 3000 - 2000 pb | ||
! rowspan="2"| - | ! rowspan="2"| - | ||
| + | |align=center| 5 | ||
| + | |align=center| 11 | ||
|- | |- | ||
|align=center|5 | |align=center|5 | ||
| - | |align=center| | + | |align=center| 5 |
| - | |align=center| | + | |align=center| 12 |
|- | |- | ||
! rowspan="1" style="background: #ccccff;"|D121 | ! rowspan="1" style="background: #ccccff;"|D121 | ||
| Line 661: | Line 505: | ||
|align=center| 3000 pb | |align=center| 3000 pb | ||
|align=center| Not digested | |align=center| Not digested | ||
| + | |align=center| 6 | ||
| + | |align=center| 3-5 | ||
|- | |- | ||
! rowspan="1" style="background: #ccccff;"|D122 | ! rowspan="1" style="background: #ccccff;"|D122 | ||
| Line 672: | Line 518: | ||
|align=center| 2500 pb | |align=center| 2500 pb | ||
|align=center| Not digested | |align=center| Not digested | ||
| - | + | |align=center| 6 | |
| + | |align=center| 6-8 | ||
|} | |} | ||
| - | + | ||
| - | + | ||
| - | + | ==> Conclusion: Most of the digestion have succeed | |
| - | + | ||
| - | + | == Extraction of the DNA == | |
| - | + | ||
| - | + | ||
| - | + | * Cutting of the parts of interest, for all the digestion that have migrated on the gels | |
| - | + | * Store of all the piece of gel O/N at -20°C. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
Latest revision as of 14:39, 31 July 2008
|
MiniPreps
DigestionDigestion Mix10µl of Miniprep (26 aug.)
Results of digestions : Electrophoresisconditions :
Extraction of the DNA
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"